Happy New Year 2026
🌟Everyone at ETAP-Lab, STROK@lliance et SYNCROSOME wishes you a Happy New Year 2026. 🌟 Toute l’équipe d’ETAP-Lab, STROK@lliance et SYNCROSOME vous souhaite une merveilleuse année 2026 !
Boost Your CNS Candidate Selection – Appealing Discount on Screening Campaigns.
To help you fast-track CNS drug discovery while optimizing your R&D budget, ETAP-Lab is launching a series time-limited screening campaigns. These exclusive opportunities give you access to our robust neuron-based assays at discounted rates, with guaranteed turnaround times and expert scientific support. Three campaigns, three opportunities:January, February & March 2026 (with limited slots per month) Ready to start a new project with us? Discover our attractive prices! #1 PROTEINOPATHY PACK Our Proteinopathy Pack is designed to assess compound
SfN NEUROSCIENCE 2025 – Empower your drug candidates with our translational preclinical models
ETAP-LAB’s team will attend the upcoming NEUROSCIENCE 2025 from November 15 to 19, in San Diego (USA), with the presentation of 2 research posters. And for the first time, we will be exhibiting to welcome you & showcase our preclinical services in the neurovascular and neurological fields. We look forward to seeing you at this World’s Largest Neuroscience Conference, organized by the Society For Neuroscience (SFN), and with a qualitative programming for the scientists coming from around the planet! 📅 When to
Pitch at the TECNIPLAST Scientific Symposium France 2025
ETAP-LAB, Preclinical CRO, pitching at the TECNIPLAST Scientific Symposium France 2025 next week! We’re pleased to announce that Sandra Robelet, Business Developer at ETAP-LAB, will be speaking at the TECNIPLAST Symposium, taking place in October 13–15, 2025 in Italy. 📌 When: October 14 🧠Topic: “The Complementarity of NAMs & Animal Models for translational AD's Drug Development”. We'll pitch about: ✔️ Challenges & Opportunities of Alternative Methods (NAMs) at preclinical stage ✔️ The "Brain-on-chip" alternative method (BIO-DIAMOND project*) in drug development stages, and how it can accelerate
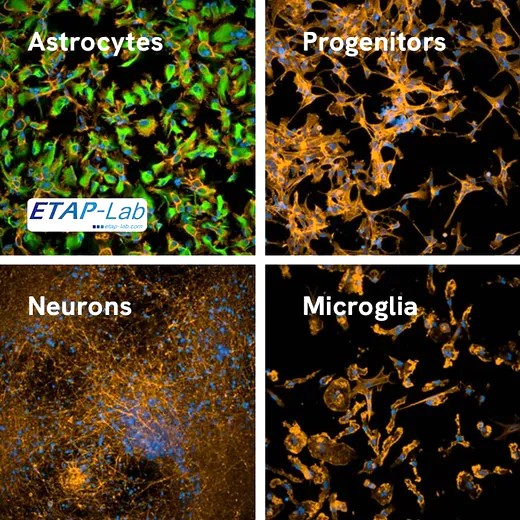
The collaboration with Revvity resulted in the launch of their staining kit, tested and approved by ETAP-LAB!
Results of the strategic collaboration between ETAP-Lab and Revvity announced We are proud to announce that ETAP-Lab, preclinical CRO expert in neuroscience, partnered with Revvity, leader in High Content Screening (HCS) solutions, to push the boundaries of discovery by combining their expertise and technology. Revvity has just launched the staining kit for astrocyte differentiation - tested and approved by ETAP-LAB - specially designed for iPSC-derived neuronal models. At ETAP-LAB, our teams contributed to this successful launch thanks to: our neuroscience expertise,
1st Anniversary of the STROK@LLIANCE building’s inauguration!
Exactly one year later
ESDR Meeting 2025 – Rediscover Our Preclinical Dermatology Expertise
Join us in Antwerp from September 10 to 13, 2025, for the annual congress of the European Society for Dermatological Research (ESDR). ETAP-Lab will be showcasing its expertise in preclinical dermatology through a dedicated booth (Booth #2) and a scientific poster session. 🔗 Learn more about the ESDR Meeting Meet Our Team at Booth #2 Our team will be pleased to present our offer of preclinical dermatological models in skin inflammation, wound healing as well as skin grafting. Schedule a meeting now with Jean-François
Neurology poster at the AFSSI Connexions 2025
Neurology poster at the AFSSI Connexions 2025 ETAP-Lab will be present at the AFSSI Connexions 2025 in Toulouse on July 1 and 2 to present the poster resulting from the research work of the laboratory dedicated to Neurology and Neurodegenerative Diseases. Healthcare R&D and Innovation: French preclinical CROs at the heart of European leadership AFSSI (the French Association of Service and Innovation Companies), in partnership with AD'OCC, the Occitanie region's economic development agency, is organizing the 12ᵉ edition of AFSSI Connexions on July
Preclinical Dermatology – Discover our Poster at SPIN 2025 Congress in Paris
Preclinical Dermatology - Discover our Poster at SPIN 2025 Congress in Paris ETAP-Lab will be presenting a poster during the next edition of the SPIN 2025 Congress, organized every 3 years by the René Touraine Foundation, focused on Skin Inflammation & Psoriasis diseases. Let's talk inflammation at SPIN with our poster on our reference models using our unique translational approach focused on assessing rodent behavior and well-being. We share an overview of over 10 years of study results, showing just how reproducible
Booth & Partnering at BIO Convention 2025 in Boston
Booth & Partnering at BIO Convention 2025 in Boston Our team will be at BIO 2025 in Boston (June 16-19), the right spot for Health Innovation! 📌 You can visit us at Booth 1935 with Eurobiomed, you'll meet Jerome Bernard and Sandra Robelet. 📆 We are partnering at BIO Convention 2025. You can connect through the BIO Partnering™ system and book a meeting to explore how we can support you in your drug development as a preclinical CRO. "As a CRO, we have to