Newsletter #11: World Alzheimer’s Day: review of the ETAP-Lab preclinical models
We'd like to take the opportunity, on World Alzheimer's Day, to revisit the models ETAP-Lab makes available to the scientific community for use in the search for new therapies to fight this disease. Building on strong neurosurgery and behavioural analysis expertise, ETAP-Lab has developed a new model of Alzheimer's disease, using human beta-amyloid peptide oligomers (hABO). Derived from a unique know-how, hABOs induce reproducible memory deficits in the elderly mouse, correlated with alterations in neurobiological markers characteristic of AD. In comparison
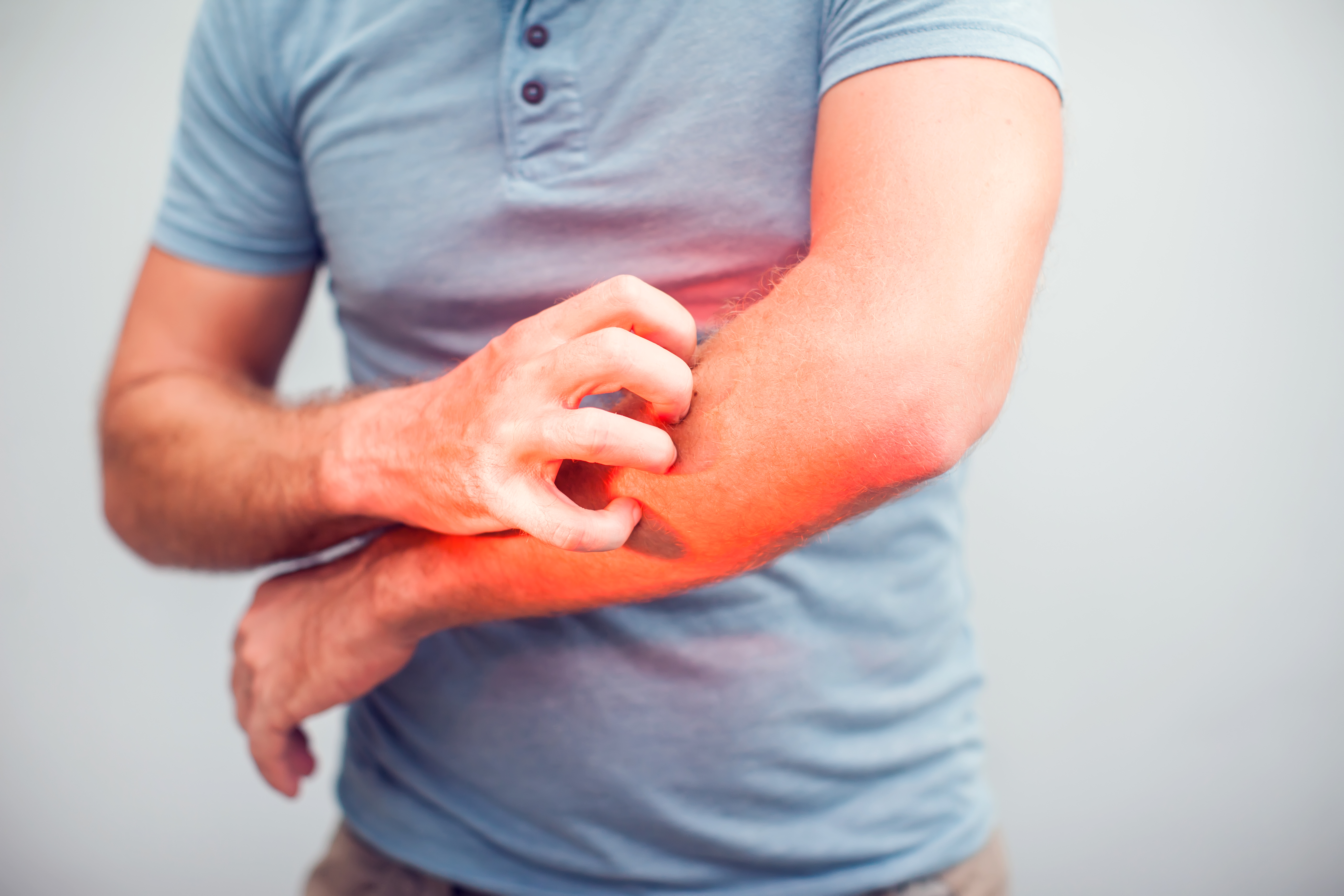
Newsletter #10 – New preclinical model of pruritus and update to our other atopic dermatitis models
Atopic dermatitis is a skin condition affecting 4% of the population worldwide. It affects all age groups, regardless of sex. Over the past 30 years, the number of diagnosed cases in industrialised countries has tripled. Characterised by dry skin and severe pruritus, this pruritic dermatosis causes major discomfort, leading to itching and even skin lesions. Other than by recourse to treatments such as the corticosteroids used to reduce skin inflammation in atopic dermatitis, pruritus management continues to be a challenging task, even
Newsletter #9 : ETAP-Lab launches its new model, associating beta-amyloid oligomers and aged mice
In May 2021, ETAP-Lab is adding its latest mouse model of Alzheimer's disease to its catalogue. Using our expertise in the manufacture of oligomers from A-beta1-42 peptide, we have demonstrated that these compounds are toxic to the central nervous systems of aged mice and cause changes in the expression of synaptic markers and apoptotic signalling, associated with a decrease in their memory capacity. ETAP-Lab has been producing A-beta1-42 peptide oligomers since 2019, and offers an in vitro testing service for the screening
Newsletter #8: The ETAP-Lab Psoriasis Model
The psoriasis model induced by the chronic application of imiquimod (IMI) proposed by ETAP-Lab reproducibly replicates the key markers of the pathology observed in humans: hyperproliferation of keratinocytes, hyperkeratosis and parakeratosis. At the systemic level, the strong activation of the immune system alongside the increased expression of characteristic cytokines such as IL-6, Il-17, IL-23 and TNF-alpha is also consistent with human pathology. Psoriasis is not, however, limited to these parameters classically measured in animals. Indeed, the skin discomfort induced by
New hope for Alzheimer’s disease: what’s combining therapeutic strategies all about?
Evidence from various studies indicates that Alzheimer's Disease (AD) develops as a result of interaction between multiple complex and overlapping pathophysiological pathways, suggesting that a pharmacological treatment with a combinatorial approach may be more beneficial than monotherapy. AD currently represents the most common form of dementia in elderly people, and with rising life expectancy the number of patients suffering from AD is expected to increase over the coming years, reaching 132 million by 2050 (World Alzheimer Report). The causes of AD are
Tau Clinical Trials – What can be learned from failure?
On the basis of the amyloid hypothesis of Alzheimer’s Disease (AD), most of the clinical trials conducted have concerned Aβ clearing therapy. Because of multiple failures in anti-amyloid clinical drug trials, more attention has been drawn to therapies targeted at tau in AD drug development. Disease-modifying therapies for AD and other tauopathies are targeting post-translational modifications, tau aggregation, microtubule stabilization, regulation of tau expression and tau clearance (immunotherapies). ClinicalTrials.gov currently lists about 120 studies targeting tau as a treatment for
6 reasons to include tau oligomers in your R&D strategy for the treatment of Alzheimer’s Disease
As its name suggests, the microtubule-associated protein tau is known mainly for its role in stabilising the microtubules, thus contributing to maintenance of the axonal transport mechanisms essential to the survival of neurons. However, the physiological roles of tau are not limited to this function alone; tau is also involved in synaptic plasticity as well as in the regulation and protection of the genome . As we shall discuss in this article, changes in the functions of tau work together
Tau oligomers: a new tool for drug discovery in AD
One common pathological hallmark of neurodegenerative disease (ND) is the aggregation and accumulation of misfolding proteins, resulting in neuronal dysfunction and brain damage. A growing body of literature suggests that amyloid oligomers (including tau) are the root cause of ND. Significant investment has been made in drug development. However, no curative molecule is currently available on the market. This may be due to several factors - most notably to flawed preclinical research in which the use and outcome of animals
Model of atopic dermatitis in mice: feedback – Newsletter #6
In January 2018, we announced the validation of a new model of atopic dermatitis in mice. Now that we have 18 months' experience of using it, here's our feedback. Back in January 2018, we announced the validation of a new model of atopic dermatitis in mice using topical applications of 2,4-Dinitrochlorobenzene (DNCB) over a period of 5 weeks in Balb/c mice (Newsletter #1). This validation allowed us to show that the model reproduces the essential characteristics of human pathology, and in
ETAP-Cell: a new in vitro pharmacology service for drug research in neurodegenerative diseases – Newsletter #5
The ETAP-Cell project continues to make progress as per the expected timeline. The whole team is delighted to announce that the in vitro laboratory is now fully equipped and already producing its first results! In late January, ETAP-Lab created ETAP-Cell, a new in vitro pharmacological service in the field of evaluating proteopathic neurodegenerative diseases using our unique know-how in the solubilization and stabilization of oligomeric proteins (AβO, αSO and TauO). Three weeks ago, we successfully produced our first batch of soluble Aβ