Tau oligomers: a new tool for drug discovery in AD
One common pathological hallmark of neurodegenerative disease (ND) is the aggregation and accumulation of misfolding proteins, resulting in neuronal dysfunction and brain damage. A growing body of literature suggests that amyloid oligomers (including tau) are the root cause of ND. Significant investment has been made in drug development. However, no curative molecule is currently available on the market. This may be due to several factors – most notably to flawed preclinical research in which the use and outcome of animals models is crucial to bridging the translational gap to the clinic. The selection of a predictive preclinical model is therefore pivotal to addressing the clinical question. We report a newly-developed in vitro AD model, induced by the highly reproducible human tau oligomer (TauO) preparations for drug screening. This model allows testing for neuroprotective compounds and/or oligomer-specific antibodies. Compound efficacy can be assessed before, concomitantly or after a TauO challenge in rodent primary cortical neuron cultures.
Contents
1. Characterization of TauO preparation
2. Neurotoxicity of TauO preparation
3. TauO induce neurotoxicity in a dose-dependent manner
1. Characterization of TauO preparation
ETAP-Lab has developed a method of producing stable misfolded TauO from recombinant full-length human tau protein (2N4R, 441 aa) without chemical modification or helper proteins. This preparation contains a mixture of trimers and low-molecular-weight oligomers, as well as remaining monomeric forms of the protein (Fig. 1).
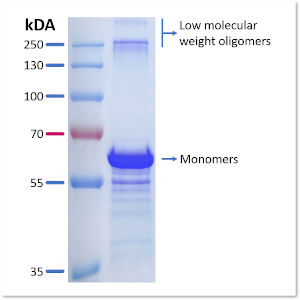
Fig.1: Characterization of TauO preparation by SDS-PAGE/Coomassie staining: SDS-page gel profile, revealed by Coomassie blue staining, demonstrating that the produced oligomers are a mixture of trimers and low-molecular-weight oligomers.
2. Neurotoxicity of TauO preparation
TauO-induced neurotoxicity was evaluated in primary rat cortical neurons. Incubation of cortical neurons with TauO decreases cell viability quite significantly (*; p<0.0001), while neuron incubation with the same concentration of monomers showed no neurotoxic effect (Fig. 2). Moreover, neuron treatment with Brain Derived Neurotrophic Factor (BDNF) significantly reversed TauO-induced neurotoxicity (#; p<0.0001).
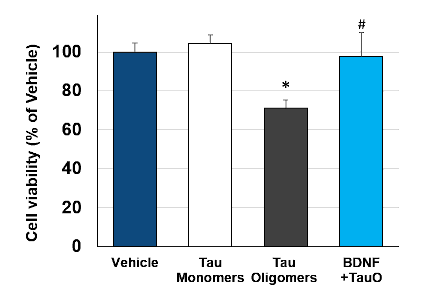
Fig.2: Neuron incubation with Tau monomers, TauO and TauO+BDNF: Incubation of neurons with TauO quite significantly decreases cell viability, while monomers showed no neurotoxic effect. TauO-induced neurotoxicity is reduced by BDNF, used as a positive control. Data are expressed as percent of vehicle (set at 100%) and represent the mean ± SD (n=12, N=3). * P<0.0001 vs. vehicle-treated cells, # P<0.0001 vs. TauO-treated cells (Scheffe’s test)
3. Dose-dependant neurotoxicity induced by TauO preparation
Primary cortical neurons, challenged with TauO over the concentration range of 0.5 to 10 μM, showed clear and biphasic dose-dependent neurotoxicity, with a clear cut-off around 9 µM (Fig. 3).
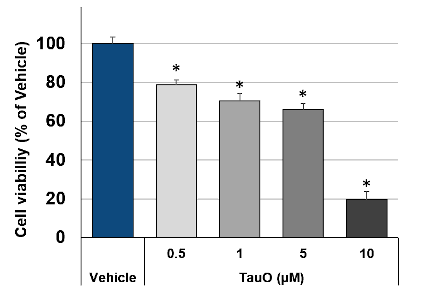
Fig.3: TauO induce dose-dependent neurotoxicity in primary cortical neurons: Neuron incubation with increasing doses of TauO showed dose-dependent neurotoxicity. Data are expressed as percent of vehicle (set at 100%) and represent the mean ± SD (n=12, N=3). * P<0.0001 vs. vehicle-treated cells (Scheffe’s test).
Primary cortical neurons were challenged with 4 independent batches of TauO to evaluate the reproducibility of the pharmacological assay. Results of cell viability showed very low batch-to-batch variabilities (Fig. 4).
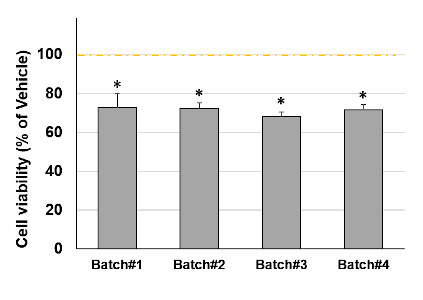
Fig.4: Alzheimer’s disease model reproducibility
Neuron incubation with different TauO batches induced a highly reproducible and significant neurotoxicity. Data are expressed as percent of vehicle (set at 100%) and represent the mean ± SD (n=3, N=1). * P<0.0001 vs. vehicle-treated cells (Scheffe’s test)
Should you have any questions or require further information, our team of neurobiologists is at your disposal: Contact
More information about our neurodegenerative diseases models
Complementary articles about tau and Tau oligomers
6 reasons to include tau oligomers in your R&D strategy for the treatment of Alzheimer’s Disease
New hope for Alzheimer’s disease: what’s combining therapeutic strategies all about?