Newsletter #3

New ETAP-Lab studies on TEMISIS’ drug candidate TEM1657 for Psoriasis
ETAP-Lab performed new studies for the account of TEMISIS on a mouse imiquimod-induced psoriasis model. The output of these studies showed that TEM1657 brings remarkable effects on psoriasis:
• Higher efficacy compared to the reference molecule
• No side effects
• Oral administration, which is new for the psoriasis treatment
The French biotech company “Temesis”, subsidiary of Plant Advanced Technologies, recently published a new press release to announce these new promising preclinical results on psoriasis with TEM1657.
The results of these new ETAP-Lab studies, together with in vitro studies, have been presented by TEMISIS in a poster at the International Investigative Dermatology 2018 in Orlando (Florida, U.S.A.) last May.
The mechanism of action of TEM1657 uses the Th17-dependant pathway which does not only give hope to psoriasis treatment but also to widespread inflammatory conditions such as Crohn’s disease or ulcerative colitis.

Figure 1: In vitro efficacy profile: microscopic view of the cells

Figure 2: In vivo efficacy profile: PASI scoring

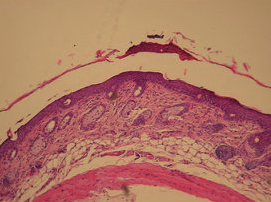
Figure 3: In vivo efficacy profile: Skin shape and histology
Flash News
Benefits of a probiotic mixture
on skin inflammation
Published in Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry,
2018 August 13th
There is a growing interest in the effects of probiotics for the prevention and treatment of skin diseases due to their immunomodulatory and anti-inflammatory properties.
In collaboration with PiLeJe Group specialized in Micronutrition and Phytotherapy, ETAP-Lab has assessed a mixture of five bacterial strains in the prevention of chronic skin inflammation in mice.
These very interesting results have shown that the probiotic mixture tested considerably limited the induced chronic skin inflammation at both macroscopic and microscopic levels. Therefore, its use could be valuable for dermatological disorders such as atopic dermatitis and psoriasis by preserving skin integrity and homeostasis.