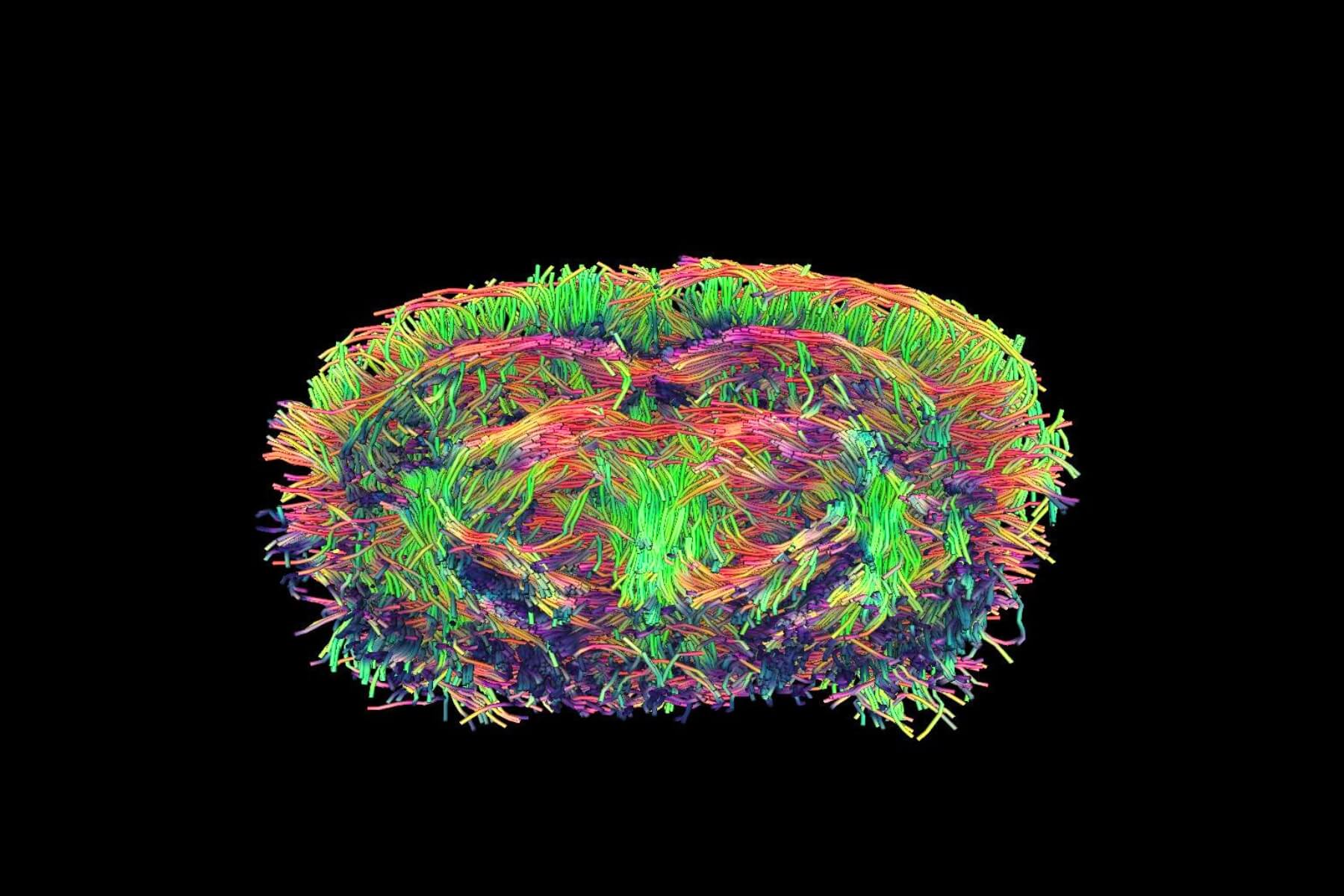
Our in vitro and in vivo models
STROK@LLIANCE offers a wide range of translational models, from small to large animals, to meet your needs in preclinical efficacy pharmacology and non‑GLP safety. We also provide in vitro tests on human blood and neuronal cultures to identify your lead compounds and support their mechanism of action. Discover our full range of services dedicated to preclinical stroke pharmacology.
A UNIQUE PARTNERSHIP
A public‑private alliance to overcome the translational roadblock of stroke.
STROK@LLIANCE is a partnership created to industrialize innovative models developed by the PhIND laboratory (UMRS 1237 INSERM/UniCaen) and make them accessible for routine studies within the only CRO with a team 100% dedicated to stroke.
Located next to the CYCERON biomedical imaging center and the BB@C institute, STROK@LLIANCE benefits from one of the most dynamic ecosystems in the world for stroke research.
More than a technology transfer, the partnership brings together a unique combination of expertise and technologies to offer the best of academic and private know‑how within a single service.
Discover our platforms dedicated to supporting your studies
Our team of experts conducts your preclinical efficacy studies in a state-of-the-art environment to ensure robust, high-quality, and repeatable results.
STROK@LLIANCE integrates cutting‑edge technologies for translational research.
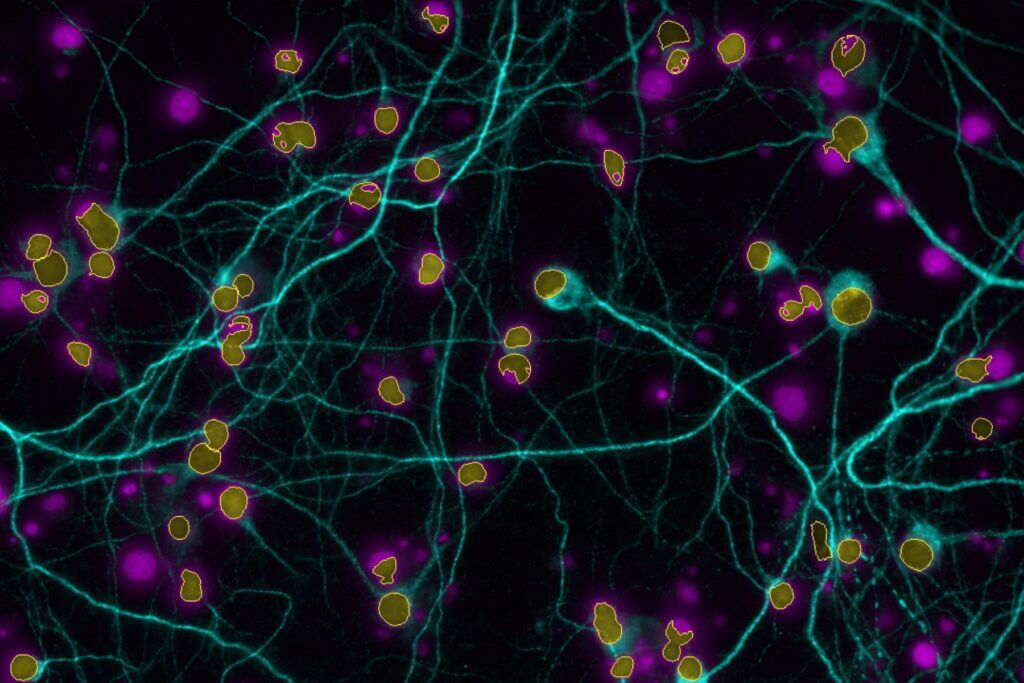
High-Content Imaging
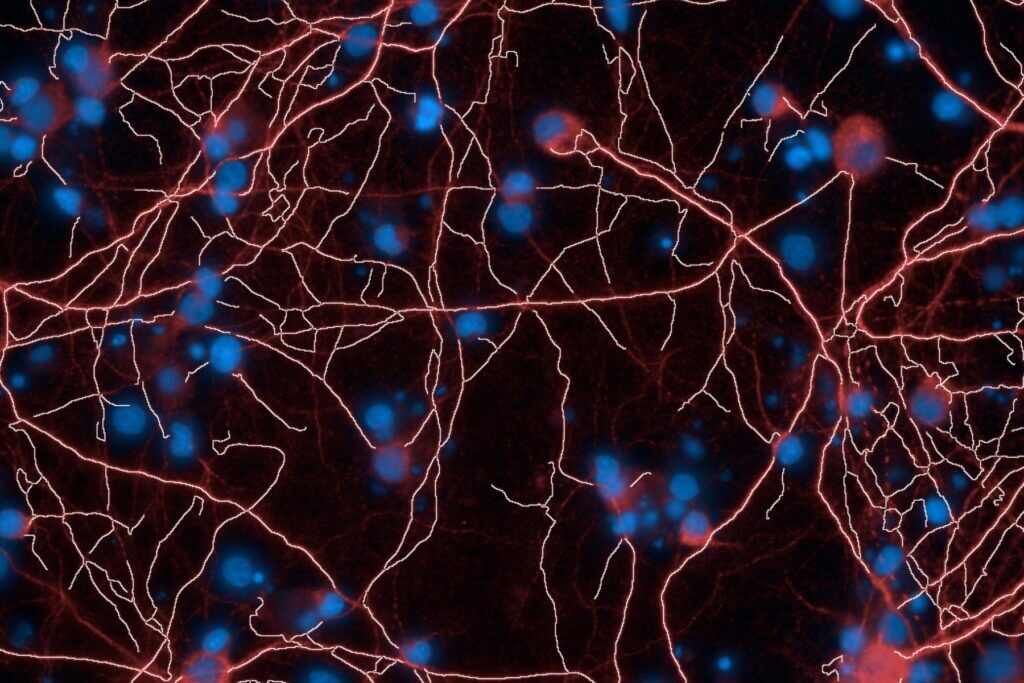
Brain‑on‑Chip platform

Small and Large Animal Facilities
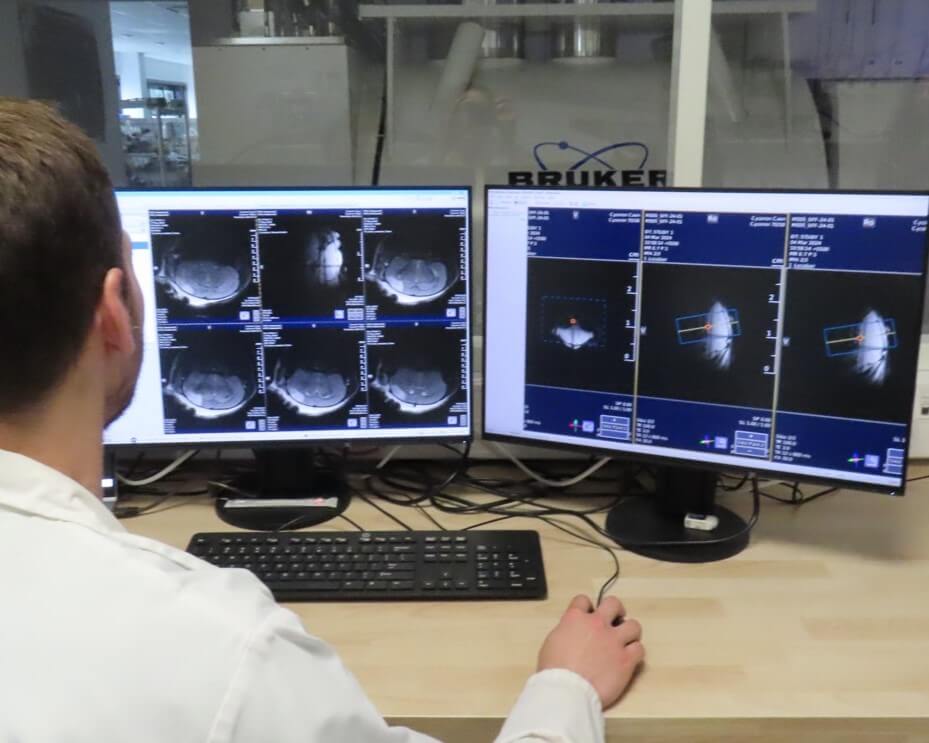
Preclinical Medical Imaging

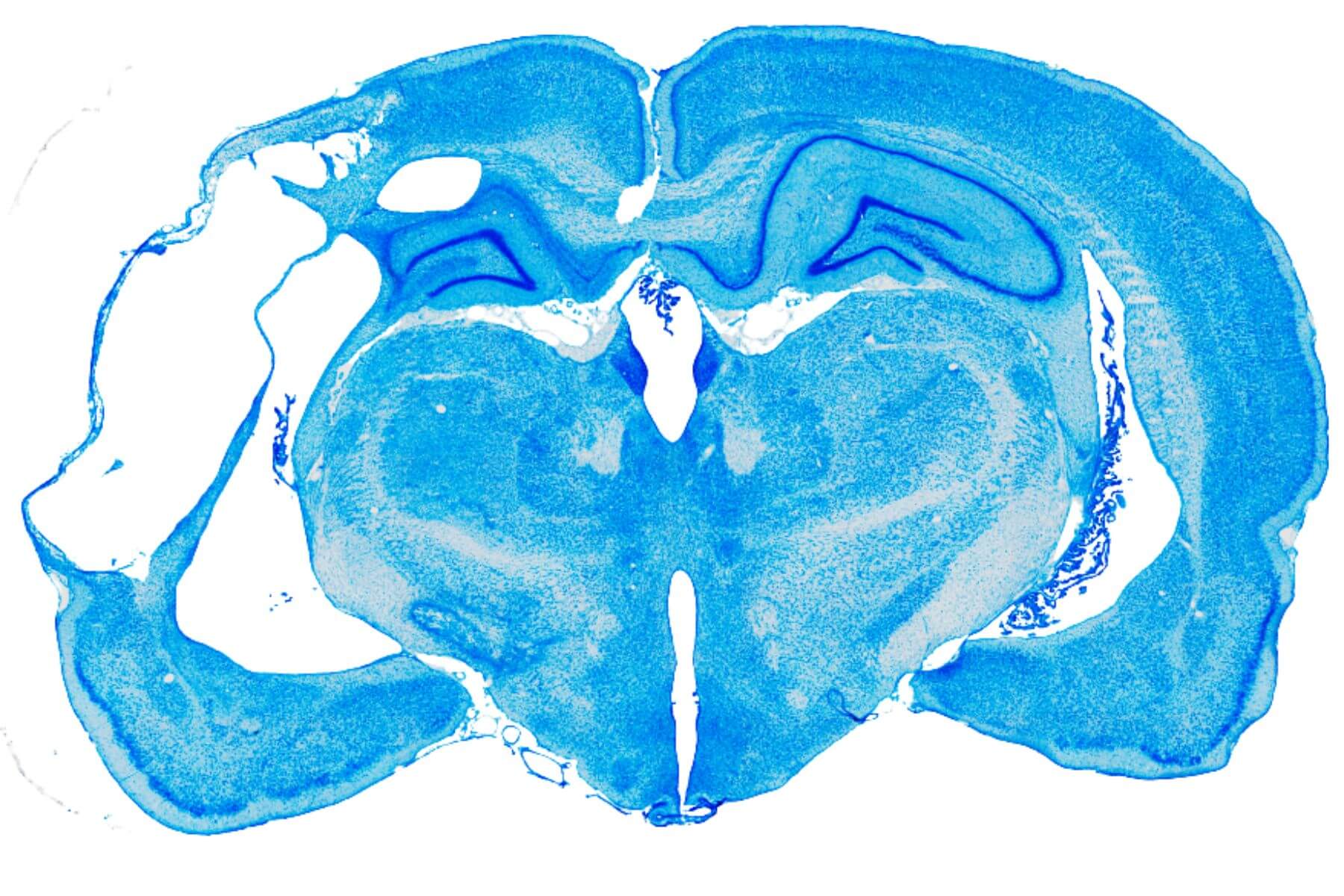
Immunohistochemistry, Immunofluorescence, and Histology
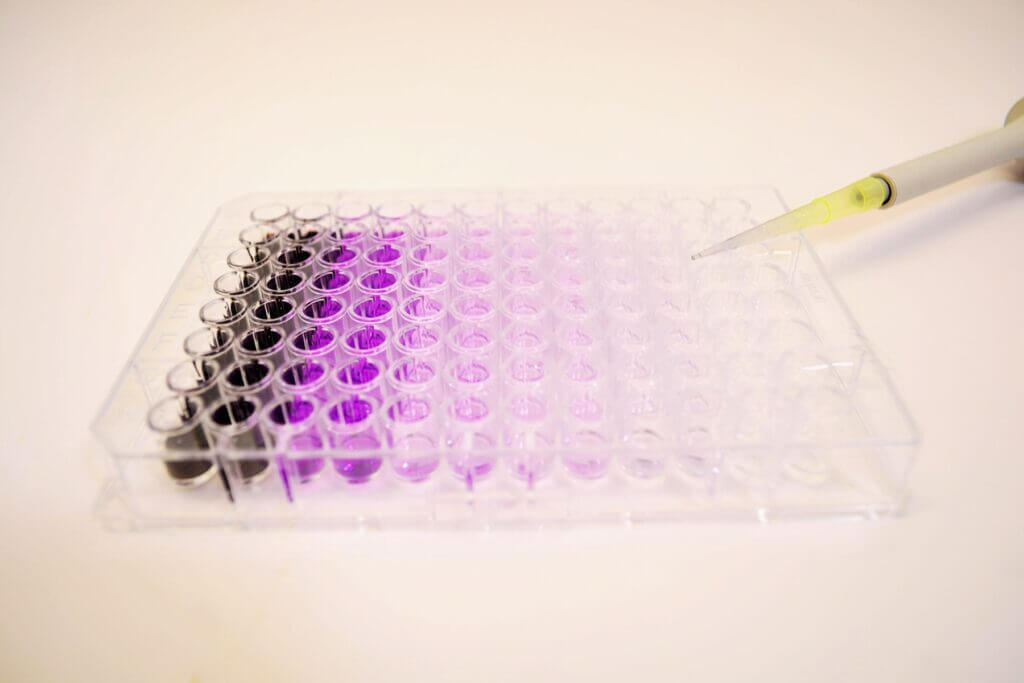
Immunoassays
Scientific resources
POSTER: “Translational Relevance and Versatility of the Thromboembolic Stroke Model in Mice: Implications for Pharmacological and Safety Evaluations”
Preclinical models of cerebral haemorrhage
Newsletter #7: From bench to bed side: how the new preclinical stroke models change the game.
Newsletter #6 – Modeling diversity in preclinical stroke
Strok@lliance – Newsletter #5
Strok@lliance – Newsletter #4
Strok@lliance – Newsletter #2
Strok@lliance – Newsletter #1
Transform your stroke-targeting innovations into therapeutic success.
STROK@LLIANCE conducts preclinical pharmacology studies dedicated to neurovascular and CNS disorders, combining CRO‑level industrial standards with the scientific expertise of a research laboratory. We offer a full range of services, from cell to large animal, including standard and customized studies: POC, PK/PD, non‑GLP safety, biomarker identification and monitoring. We address all your needs in preclinical evaluation of neurovascular and hemostasis‑related conditions.
We have four areas of expertise in stroke and acute CNS injury: in vitro screening on human blood, ischemic stroke, hemorrhagic stroke, and bleeding‑risk evaluation for therapies.
Our Brands
STROK@LLIANCE
Founded 35 years ago with recognized expertise in neurology, ETAP‑LAB has expanded into additional therapeutic areas. Since 2017, STROK@LLIANCE has represented our neurovascular expertise, built in partnership with UniCaen.
A SOCIETAL CHALLENGE
STROK@LLIANCE contributes to the development of tomorrow’s therapies.
Stroke, the third leading cause of mortality and acquired disability worldwide, remains a major global health challenge. With 94 million people affected and 160 million DALYs in 2021, it still lacks truly effective therapeutic solutions.
STROK@LLIANCE provides next‑generation in vitro and in vivo models, combined with advanced imaging and analytical technologies, to accelerate identification and validation of a ground-breaking treatment for pharmacological management of stroke.