A range of services to accelerate your drug pipeline
Contact an expertJust like many others before you, you can accelerate and secure your drug pipeline with ETAP-LAB’s expert preclinical pharmacology services. Our premium, integrated services combine the industrial standards of a CRO with the scientific expertise of a research laboratory to effectively evaluate the efficacy of your therapeutic innovations.
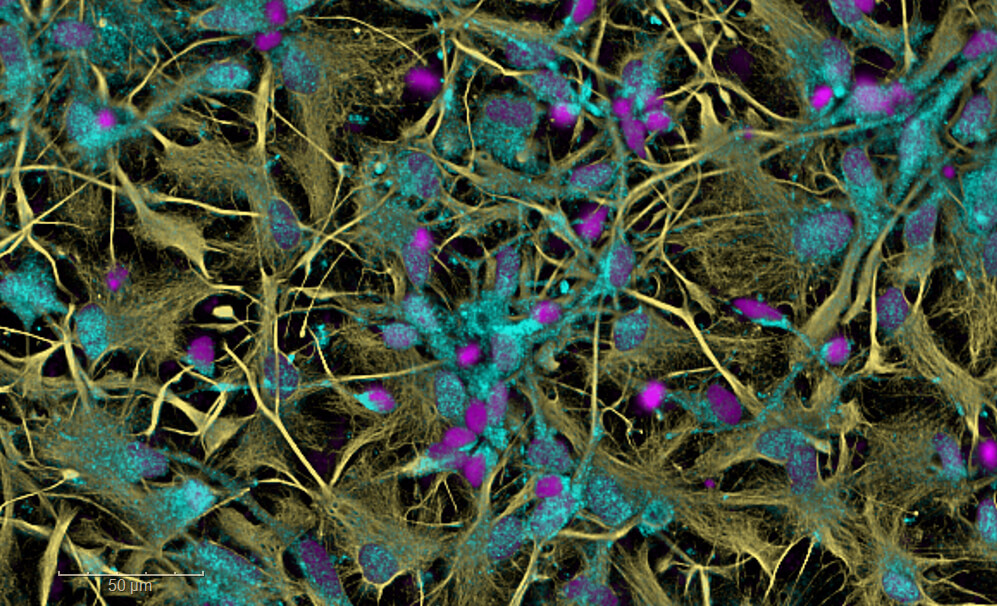
In vitro efficacy studies
ETAP-LAB provides a range of in vitro services in neurology, neurovascular disease and hemostasis, for medium-throughput screening of your drugs in the lead optimization phase. Our cell culture laboratory uses models of neurodegenerative diseases or stroke based on primary cultures of neurons or human IPSCs in 2D. Explore complex mechanisms of action with our brain-on-a-chip models combined with high-content imaging. Our hematology laboratory will evaluate your thrombolytics, anticoagulants, or hemostatic agents on whole human blood or its derivatives.
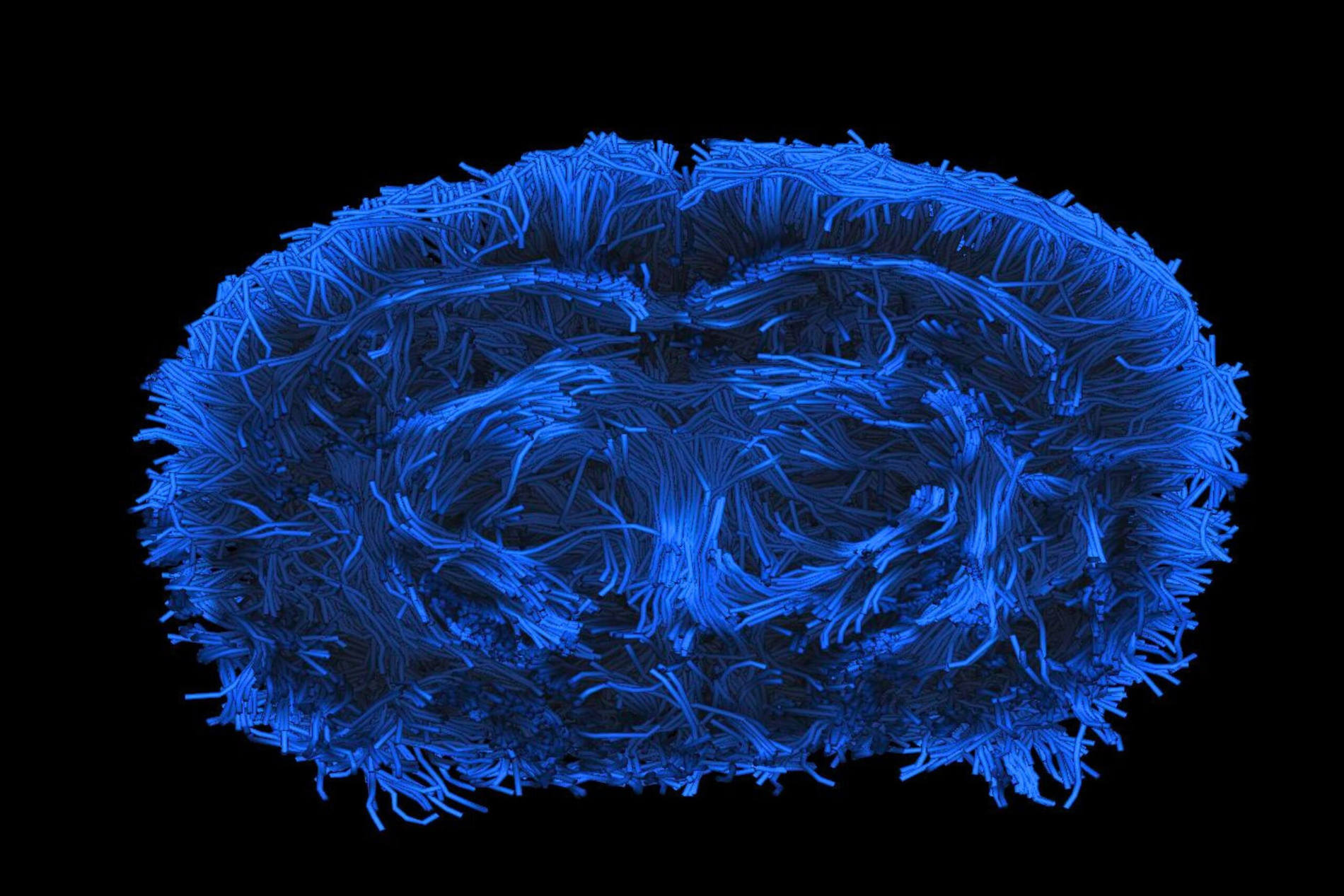
In vivo efficacy studies
ETAP-LAB’s portfolio includes gold-standard and innovative rodent models designed to accurately reproduce key pathophysiological mechanisms of target human diseases. These well-characterized and validated models are versatile and can be adapted to evaluate small molecules, cell therapies, gene therapies, biomimetics, and medical devices. ETAP-LAB can also conduct custom-designed studies in pigs.
With translational readouts (medical imaging, behavior, histology) and high standards of implementation, our studies will support your regulatory dossiers, win over medical experts, and enhance your product’s value among investors.
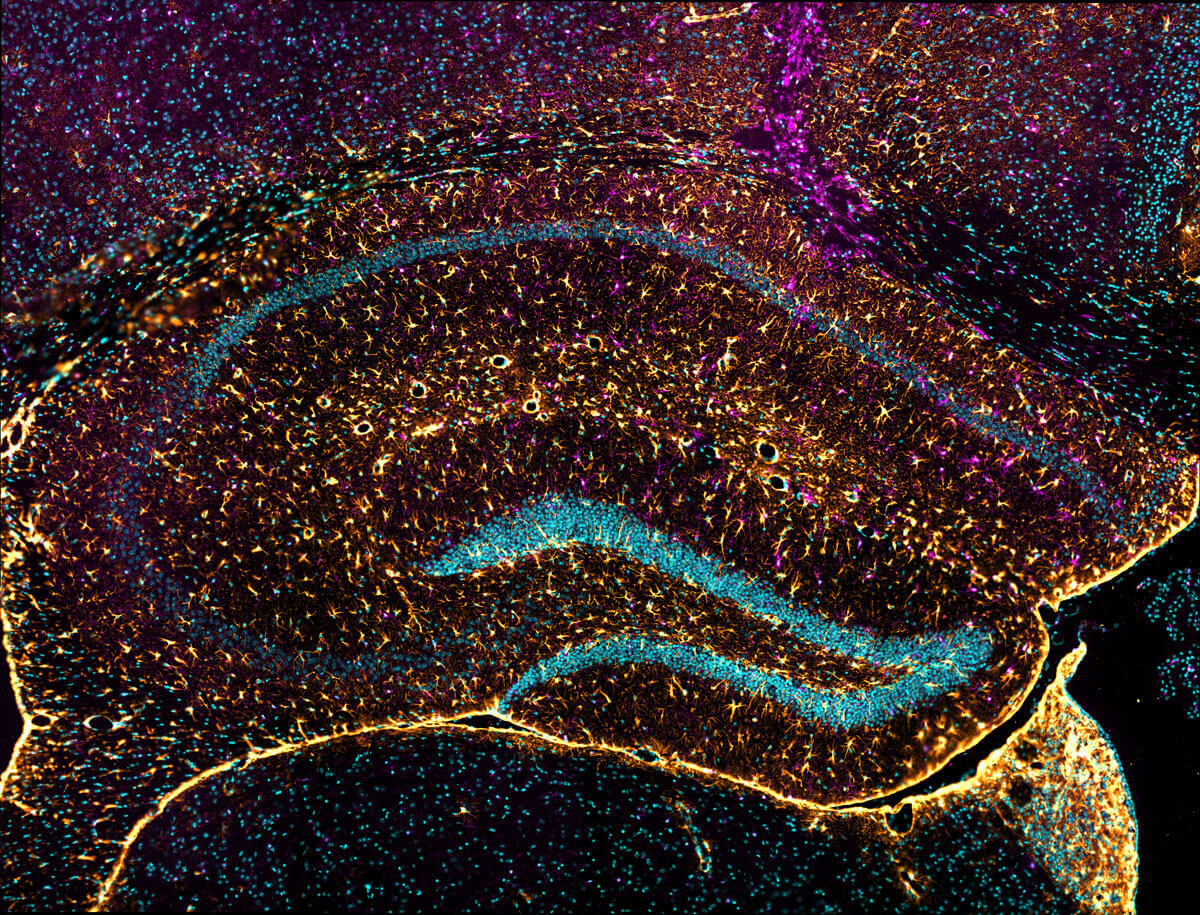
Analytical services
Finding a trusted provider to analyze specific and rare biological targets is a major challenge for pharmacologists. ETAP-LAB provides an analysis service and performs customized assays and immunohistological studies in its fields of expertise.
Leave nothing to chance with your most valued samples!

Non-regulatory safety
ETAP-LAB has developed specialist expertise in assessing the safety of drugs in the neurovascular field and has helped obtain INDs for numerous molecules in this field. Risk of hemorrhagic transformation, risk of intracranial and peripheral hemorrhage, pharmacological interactions with fibrinolytic or antithrombotic treatments… Discover our range of non-regulatory safety studies and talk to our experts who will help you anticipate questions from drug evaluation agencies.

PK-PD
All of our in vivo models are compatible with PK-PD studies. We can also conduct neuro-PK studies in rodents to evaluate the bioavailability of your compound or innovative galenic formulation in the brain.
We have established a network of trusted partners to develop analytical methods and perform assays of your compound in the blood and tissues of interest.

Scientific consulting
Besides conducting preclinical studies, ETAP-LAB supports and advises its customers in order to select the most appropriate models and identify the protocols best suited to their therapeutic strategy.
In addition, ETAP-LAB works with a network of scientific KOLs who can provide their expertise to answer questions about a specific target or mechanism.
Finally, ETAP-LAB will help you promote your research (publications, posters, oral presentations), produce technical and scientific responses to drug evaluation agencies, and evaluate independently obtained results.
We combine scientific expertise with the application of strict industry standards to effectively realize your preclinical projects.
ETAP-LAB is a preclinical CRO specializing in neurology, cardiology, neurovascular medicine, and dermatology.
Haven’t found the service you’re looking for? Please contact us. We can take you through our catalog or tailor our offering to your specific needs.





