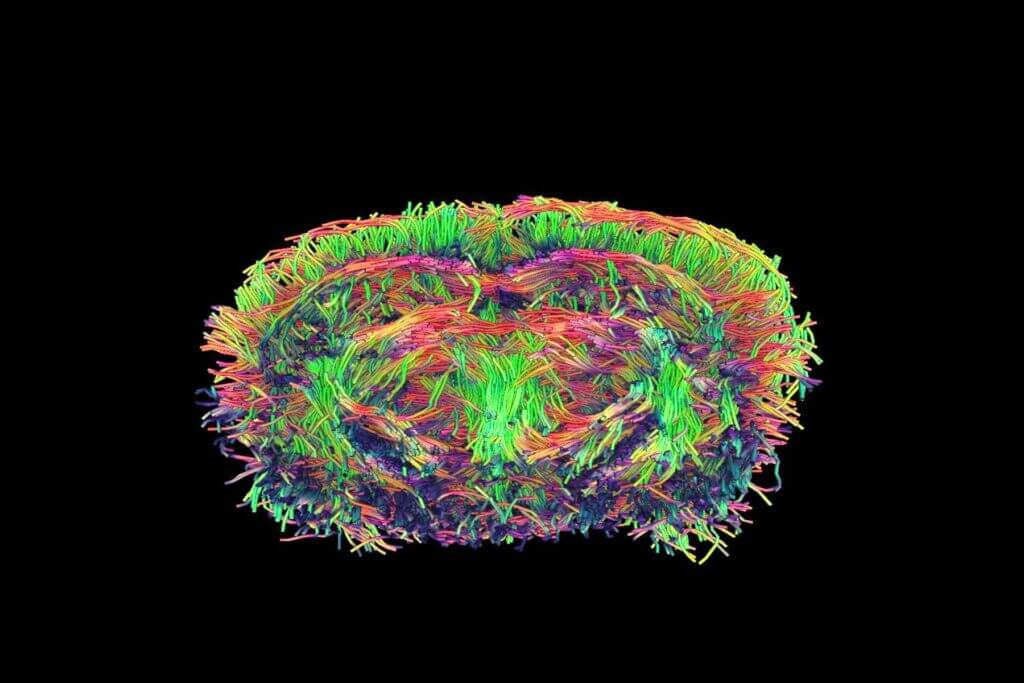
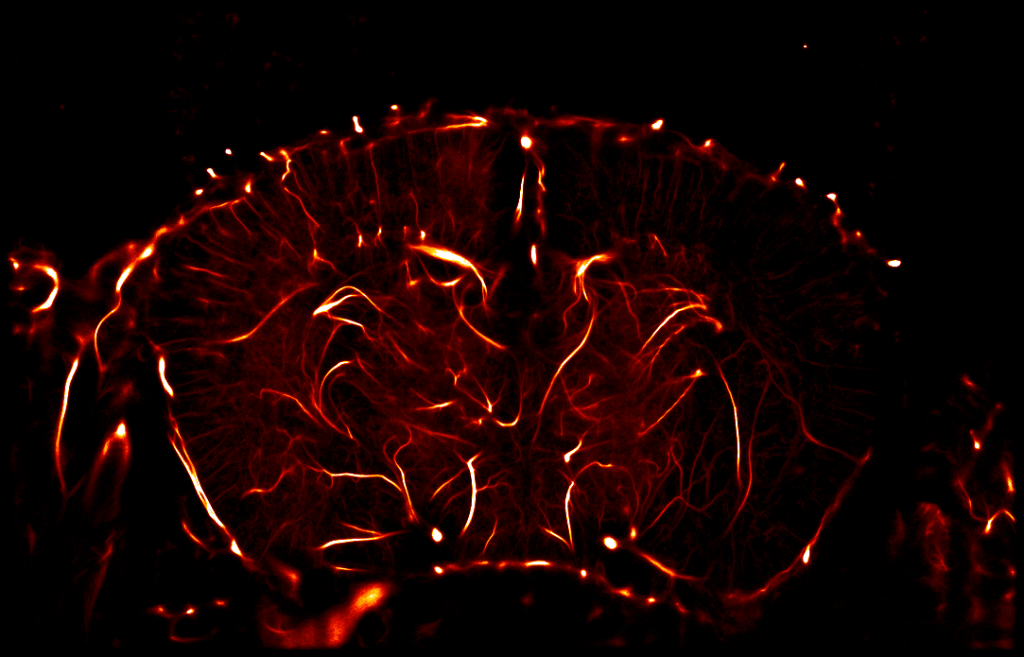
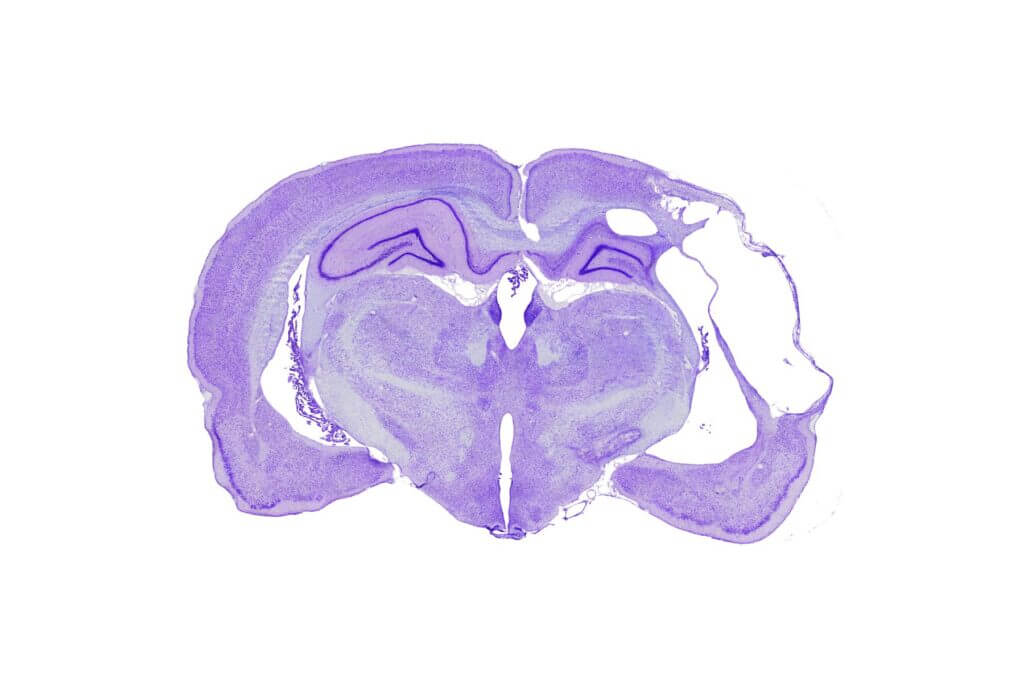
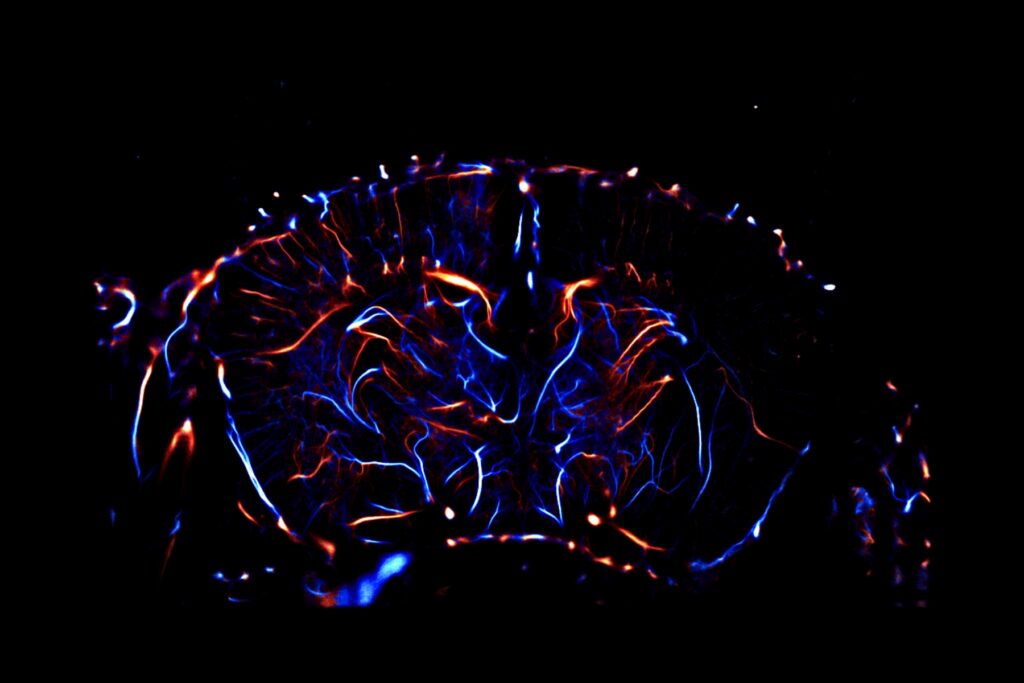
in vivo pharmacology models in Neurovascular & Stroke

STROK@LLIANCE, offers you the world’s most innovative translational portfolio of animal models of stroke, implemented by a highly experienced team of experts 100% dedicated to preclinical stroke research.
Your one‑stop shop for pharmacological studies on stroke: the complementarity of our stroke animal models enables compliance with all STAIR recommendations and regulatory agency requirements within a single CRO.
Thanks to our privileged partnership with a world‑renowned academic team, our protocols continuously evolve to integrate the latest scientific advances in all project phases.
The efficacy of your molecules can be assessed using a set of complementary and multi‑scale approaches: anatomical and molecular medical imaging, neurofunctional imaging, behavioral monitoring, immunohistochemical analyzes, and molecular biology. Every parameter of the study design – administration route, timing, therapeutic regimen, readouts, group size, and inclusion of reference compounds – is customizable and carefully harmonized to meet your specific needs.
Efficacy animal models
Hemorrhagic stroke models (ICH)
Intracerebral hemorrhagic strokes (ICH) are characterized by an accumulation of blood within the brain parenchyma following the rupture of a blood vessel, leading to a cascade of inflammatory and neurotoxic events. Rely on our validated and wellestablished ICH animal models to accelerate the selection of your drug candidates by assessing their efficacy and strengthening your proofofconcept (POC). Our ICH models are also suitable for safety studies (bleeding risk) or pharmacological interaction studies (bleeding potentiation or antidotes).
ICH induced by collagenase injection
Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
ICH induced by autologous blood injection
Stereotaxic infusion of a predetermined volume of autologous blood into the striatum generates a reproducible hematoma, inducing increased intracranial pressure, brain toxicity, and an inflammatory response associated with clear neurological deficits. This model provides a reliable tool for evaluating the efficacy of your drug candidates on edema, inflammation, or neuroprotection, while eliminating potential biases linked to their effects on bleeding volume.
Stereotaxic infusion of a predetermined volume of autologous blood into the striatum generates a reproducible hematoma, inducing increased intracranial pressure, cerebral toxicity, and an inflammatory response associated with clear neurological deficits. This model is a reproducible tool for evaluating the efficacy of your molecules on edema, inflammation, or neuroprotection, eliminating potential biases related to their effect on bleeding volume.
Validated model available in rats.
Distal ischemic stroke models
Our distal ischemic stroke models are induced by thrombotic occlusion of the M2 branch of the middle cerebral artery (MCA). They generate a focal cortical lesion whose characterization has demonstrated strong translational value in the acute phase of stroke, compared with mechanical or chemical stroke models. Our distal ischemic stroke models are available in different versions depending on the nature of the thrombi (fibrinrich vs. plateletrich) or the presence of a comorbidity.
These models are highly relevant for your POC, PK/PD, superiority efficacy studies, combination therapy studies, pharmacological interaction assessments, and assessment of hemorrhagic transformation risk. Designed for evaluating therapeutic strategies targeting thrombi or neuroprotection, several reference compounds are available.
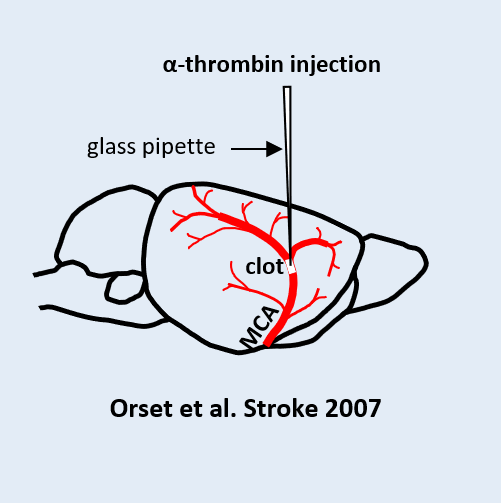
MCA occlusion by thrombin injection
This preclinical ischemic stroke model is induced by the formation of a fibrinrich clot in the MCA. This clot is efficiently lysed by standard clinical fibrinolytics (Alteplase, Tenecteplase), reflecting the patient subgroup that responds to standard-of-care fibrinolytics. This makes the model a key translational tool for assessing the efficacy of your thrombolytic candidates. This model is also highly relevant for evaluating neuroprotective compounds in an experimental setting that closely mimics the clinical reality of the acute phase of stroke (with the presence of a thrombus and pharmacological recanalization achieved within a clinically relevant treatment window). It additionally enables the study of interactions with reference fibrinolytic treatments.
Validated model available in mice.
MCA occlusion by FeCl₃ or AlCl₃ application
This preclinical ischemic stroke model is induced by the formation of a platelet‑rich clot that is resistant to standard fibrinolytics (Alteplase, Tenecteplase) but responsive to other thrombolytic agents such as N‑acetylcysteine. By reflecting the subpopulation of patients who fail to respond to standard‑of‑care treatments, this translational model is a powerful tool for assessing the efficacy and superiority of your candidates, combination therapies, and pharmacological interactions.
Validated model available in mice.
MCA occlusion by thrombin injection in diabetic animals
Proximal ischemic stroke models (MCAO)
Proximal ischemic strokes are reproduced through occlusion of the middle cerebral artery (MCAO) at its initial segments. The induced lesion encompasses extensive cortical and subcortical areas, leading to persistent sensorimotor and cognitive deficits and incomplete functional recovery. Use our MCAO models to evaluate the effects of your therapeutic strategy on longterm functional recovery, in alignment with STAIR recommendations.
tMCAO model – Mechanical ischemia/reperfusion
This preclinical ischemic stroke model relies on a transient mechanical occlusion of the MCA (tMCAO), reproducing cerebral ischemia followed by controlled reperfusion. As a historical and extensively documented model, it produces both cortical and subcortical lesions, enabling the evaluation of a wide range of therapeutic strategies against a pharmacological reference, as well as detailed exploration of their effects on tissue evolution and on longterm sensorimotor and cognitive recovery.
Validated model available in mice and rats.
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
Validated model available in mice and rats.
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
ICH induced by collagenase injection Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Validated model available in rats
Bleeding risk assessment
MCA occlusion by thrombin injection in diabetic animals
Tail bleeding assay
The tail bleeding test is a goldstandard assay used to evaluate hemostasis and platelet function in vivo.
This model is sensitive to anticoagulant and fibrinolytic treatments commonly administered in the clinic, such as heparin, direct oral anticoagulants, or Alteplase. This model supports the evaluation of candidate efficacy across multiple drug classes, such as anticoagulants, procoagulants, and antiplatelet agents.
Validated model available in mice
ICH induced by collagenase injection
Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Drug interactions
MCA occlusion by thrombin injection
This preclinical ischemic stroke model is induced by the formation of a fibrinrich clot in the MCA. This clot is efficiently lysed by standard clinical fibrinolytics (Alteplase, Tenecteplase), reflecting the patient subgroup that responds to standard-of-care fibrinolytics. This makes the model a key translational tool for assessing the efficacy of your thrombolytic candidates. This model is also highly relevant for evaluating neuroprotective compounds in an experimental setting that closely mimics the clinical reality of the acute phase of stroke (with the presence of a thrombus and pharmacological recanalization achieved within a clinically relevant treatment window). It additionally enables the study of interactions with reference fibrinolytic treatments.
Validated model available in mice.
MCA occlusion by FeCl₃ or AlCl₃ application
This preclinical ischemic stroke model is induced by the formation of a platelet‑rich clot that is resistant to standard fibrinolytics (Alteplase, Tenecteplase) but responsive to other thrombolytic agents such as N‑acetylcysteine. By reflecting the subpopulation of patients who fail to respond to standard‑of‑care treatments, this translational model is a powerful tool for assessing the efficacy and superiority of your candidates, combination therapies, and pharmacological interactions.
Validated model available in mice.
MCA occlusion by thrombin injection in diabetic animals
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
ICH induced by collagenase injection Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Validated model available in rats
ICH induced by collagenase injection
Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
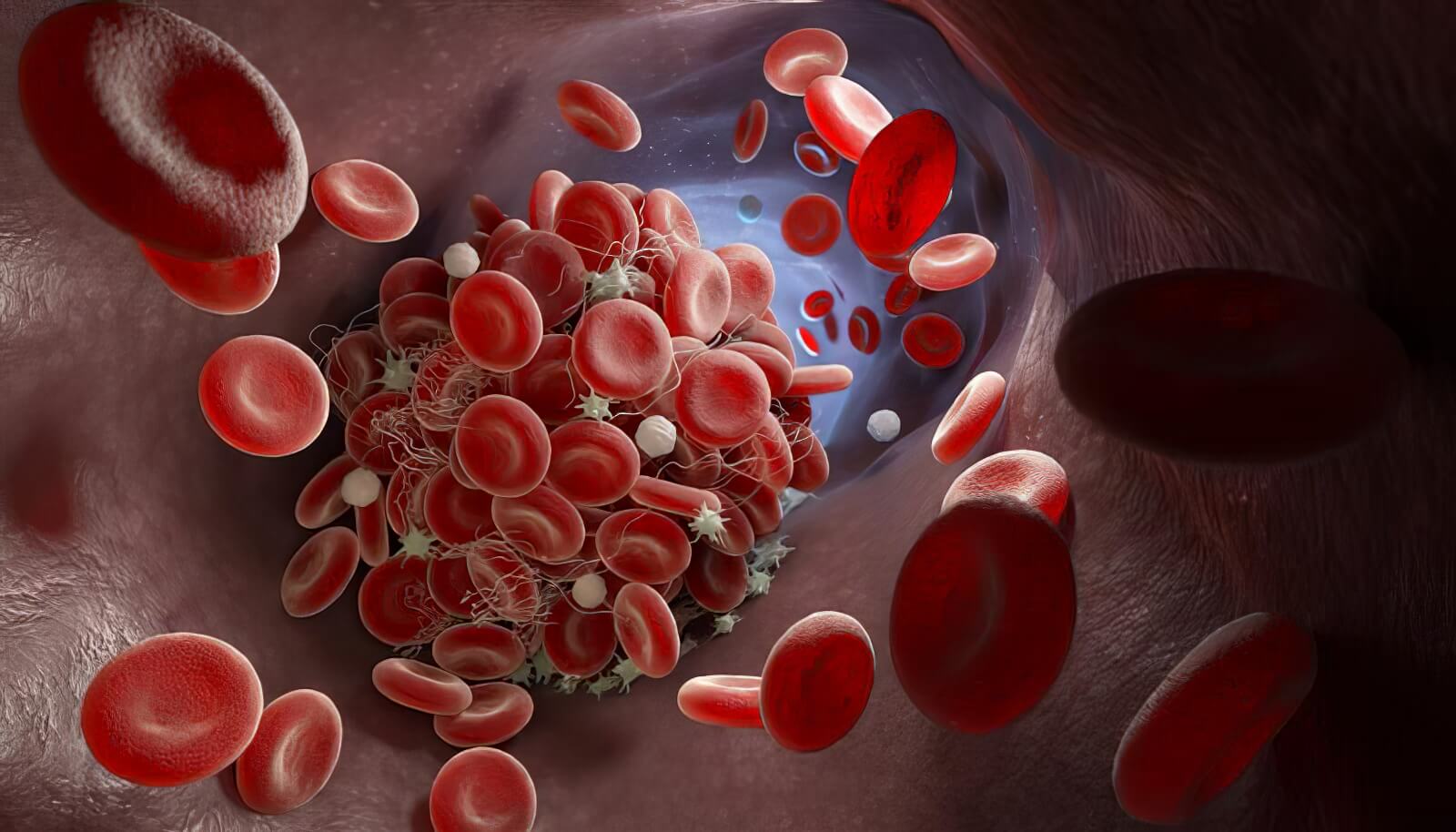
Hemostasis and blood coagulation
Tail bleeding assay
The tail bleeding test is a goldstandard assay used to evaluate hemostasis and platelet function in vivo.
This model is sensitive to anticoagulant and fibrinolytic treatments commonly administered in the clinic, such as heparin, direct oral anticoagulants, or Alteplase. This model supports the evaluation of candidate efficacy across multiple drug classes, such as anticoagulants, procoagulants, and antiplatelet agents.
Validated model available in mice
Fibrinolytics and thrombolytics
Distal ischemic stroke models
Our distal ischemic stroke models are induced by thrombotic occlusion of the M2 branch of the middle cerebral artery (MCA). They generate a focal cortical lesion whose characterization has demonstrated strong translational value in the acute phase of stroke, compared with mechanical or chemical stroke models. Our distal ischemic stroke models are available in different versions depending on the nature of the thrombi (fibrinrich vs. plateletrich) or the presence of a comorbidity.
These models are highly relevant for your POC, PK/PD, superiority efficacy studies, combination therapy studies, pharmacological interaction assessments, and assessment of hemorrhagic transformation risk. Designed for evaluating therapeutic strategies targeting thrombi or neuroprotection, several reference compounds are available.
MCA occlusion by thrombin injection
This preclinical ischemic stroke model is induced by the formation of a fibrinrich clot in the MCA. This clot is efficiently lysed by standard clinical fibrinolytics (Alteplase, Tenecteplase), reflecting the patient subgroup that responds to standard-of-care fibrinolytics. This makes the model a key translational tool for assessing the efficacy of your thrombolytic candidates. This model is also highly relevant for evaluating neuroprotective compounds in an experimental setting that closely mimics the clinical reality of the acute phase of stroke (with the presence of a thrombus and pharmacological recanalization achieved within a clinically relevant treatment window). It additionally enables the study of interactions with reference fibrinolytic treatments.
Validated model available in mice.
MCA occlusion by FeCl₃ or AlCl₃ application
This preclinical ischemic stroke model is induced by the formation of a platelet‑rich clot that is resistant to standard fibrinolytics (Alteplase, Tenecteplase) but responsive to other thrombolytic agents such as N‑acetylcysteine. By reflecting the subpopulation of patients who fail to respond to standard‑of‑care treatments, this translational model is a powerful tool for assessing the efficacy and superiority of your candidates, combination therapies, and pharmacological interactions.
Validated model available in mice.
MCA occlusion by thrombin injection in diabetic animals
Proximal ischemic stroke models (MCAO)
Proximal ischemic strokes are reproduced through occlusion of the middle cerebral artery (MCAO) at its initial segments. The induced lesion encompasses extensive cortical and subcortical areas, leading to persistent sensorimotor and cognitive deficits and incomplete functional recovery. Use our MCAO models to evaluate the effects of your therapeutic strategy on longterm functional recovery, in alignment with STAIR recommendations.
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
Validated model available in mice and rats.
Pulmonary embolism
Pulmonary embolism model induced by injection of autologous clots
The pulmonary embolism model induced by injection of autologous clots relies on preparing and injecting pre‑formed calibrated clots into the jugular vein to selectively obstruct the pulmonary arteries. This model reproduces the acute increase in pulmonary pressure observed in human clinical settings, along with significant stress on the right ventricle and secondary vascular remodeling. The severity of the condition can be adjusted based on the amount of thrombotic material used. Its strong reproducibility and rapid implementation make it suitable for quickly evaluating the efficacy of thrombolytic, vasodilatory, or cardioprotective treatments.
Rat model.
Neuroprotection and functional recovery
Proximal ischemic stroke models (MCAO)
Proximal ischemic strokes are reproduced through occlusion of the middle cerebral artery (MCAO) at its initial segments. The induced lesion encompasses extensive cortical and subcortical areas, leading to persistent sensorimotor and cognitive deficits and incomplete functional recovery. Use our MCAO models to evaluate the effects of your therapeutic strategy on longterm functional recovery, in alignment with STAIR recommendations.
tMCAO model – Mechanical ischemia/reperfusion
This preclinical ischemic stroke model relies on a transient mechanical occlusion of the MCA (tMCAO), reproducing cerebral ischemia followed by controlled reperfusion. As a historical and extensively documented model, it produces both cortical and subcortical lesions, enabling the evaluation of a wide range of therapeutic strategies against a pharmacological reference, as well as detailed exploration of their effects on tissue evolution and on longterm sensorimotor and cognitive recovery.
Validated model available in mice and rats.
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
ICH induced by collagenase injection Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Validated model available in rats
Pharmacokinetics (PK) and neuro-PK
Our pharmacokinetic studies are based on flexible kinetic models tailored to your drug candidate. We analyze standard biological matrices (plasma, serum, CSF) as well as key organs or anatomical regions to accurately characterize your molecule’s tissue exposure. These robust and customizable approaches provide a solid foundation for your preclinical decisions.
PK model
We offer studies tailored to your PK and Neuro-PK needs, covering various administration routes (subcutaneous, intraperitoneal, intravenous, oral, intranasal, etc.) in both acute and chronic treatment paradigms. The bioavailability of your molecules is quantified across different compartments—including blood, brain, cerebrospinal fluid, or any other tissue of interest. This approach provides reliable data to support decision-making and accelerate the development of your therapeutic candidates.
Available in mice, rats, and pigs.
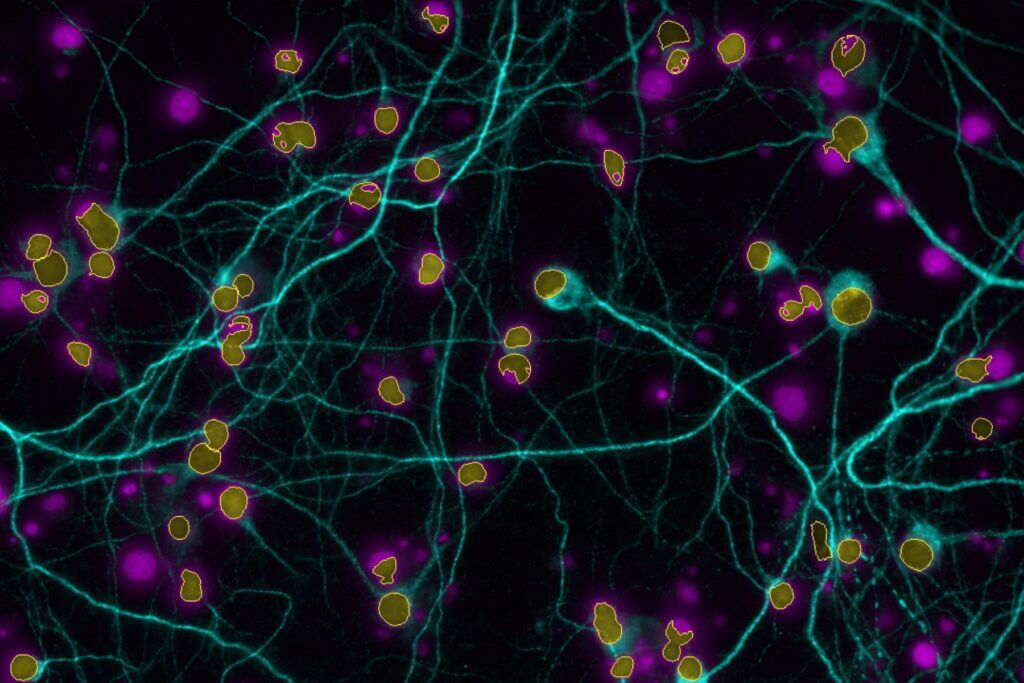
High-Content Imaging
Brain‑on‑Chip platform

Small and Large Animal Facilities
Preclinical Medical Imaging

Immunohistochemistry, Immunofluorescence, and Histology
Immunoassays
The effects of your molecules can be evaluated through complementary and multi-scale approaches.