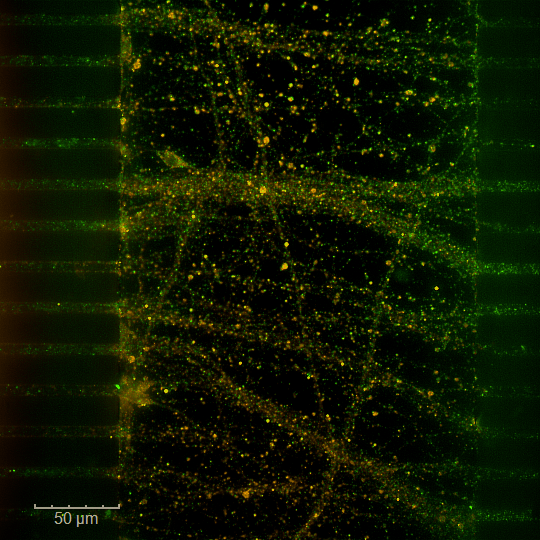
in vitro pharmacology models in Neurovascular & Stroke
With STROK@LLIANCE, accelerate the development of your ischemic and hemorrhagic stroke therapies with our preclinical in vitro models based on primary neuronal cultures and human blood-based assays. We support you in:
- Assessing the efficacy and dose-response profiles of your drug benchmarked against relevant reference compound
- Screening and identifying neuroprotective, thrombolytic, anticoagulant, hemostatic, and antidote candidates targeting bleeding events associated with hemostasis-modulating therapies
- Characterizing mechanisms of action using human whole blood, plasma, or serum for hemostasis-related investigations, or primary neuronal cultures, hIPSC-derived systems, or organ-on-chip platforms for neuroprotection studies
- Investigating drug-drug interactions between your candidate and current standard-of-care treatments used in clinical practice, for potentiation assessments or non-regulatory safety evaluations.
Fibrinolysis and hemostasis screening
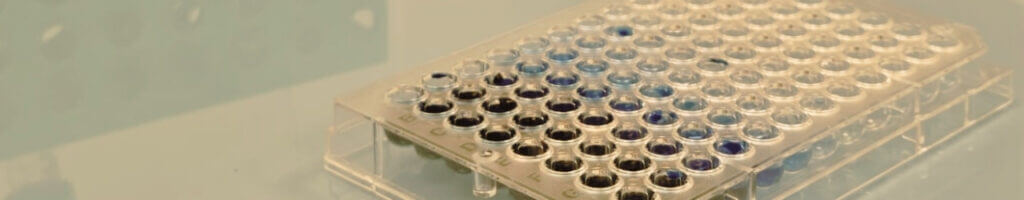
Clot lysis assay
The clot lysis assay evaluates the efficacy of your fibrinolytic candidates on human plasma pools. Performed by turbidimetry in 96-well plates, it provides rapid assessment of compounds activity through parameters such as the 50% clot lysis time (LT50). The high reproducibility of the assay ensures reliable and straightforward comparison of dose-response curves throughout lead-optimisation phases.
This assay also allows investigation of compound interactions when combined with reference fibrinolytics (Alteplase, Tenecteplase).
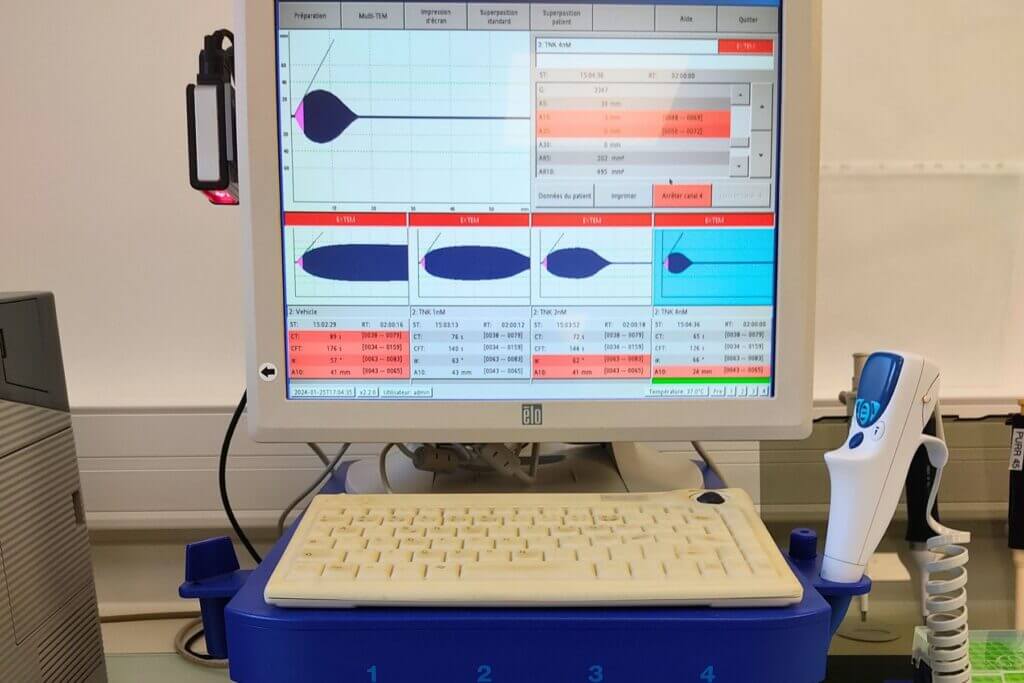
ROTEM assay
The ROTEM assay, performed on fresh human whole blood, measures the viscoelastic properties of the clot throughout its formation and subsequent lysis. A wide range of parameters can be extracted from the thromboelastogram, including the clotting time, the maximum clot firmness, and the 50% clot lysis time (LT50). This low-throughput assay offers an extensive and customisable set of hemostatic conditions for evaluating anticoagulant or thrombolytic compounds, including extrinsic or intrinsic activation pathways, as well as the addition of coagulation factors or modulators.
This assay also allows investigation of compound interactions when combined with reference fibrinolytics (Alteplase, Tenecteplase).
Neuroprotective screening
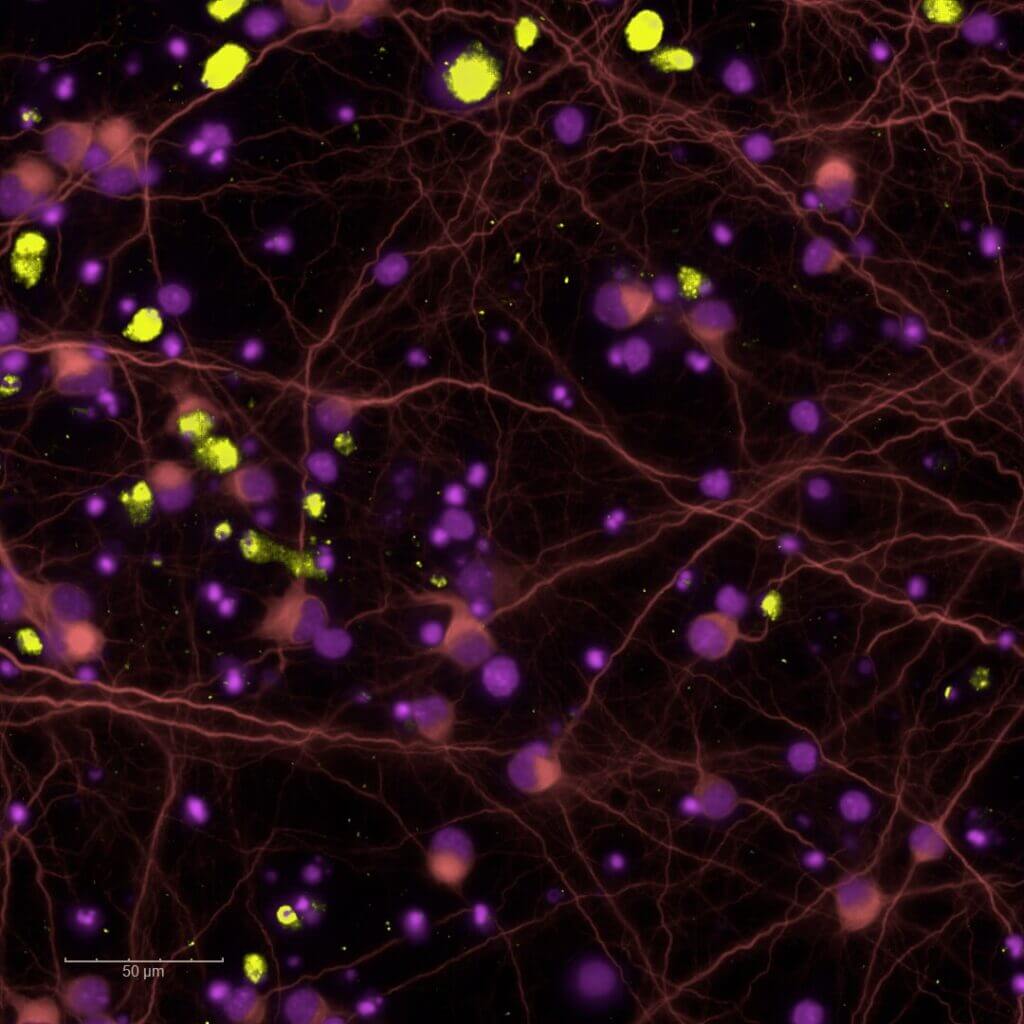
Glutamate-induced toxicity : 2D culture
Our 2D excitotoxicity model provides a rapid and reliable platform to screen neuroprotective compounds in primary rodent neuronal cultures exposed to glutamate overload, with MK801 as a reference compound.
Our high‑content imaging platform supports medium‑throughput screening of glutamate‑induced effects at both acute excitotoxic concentrations and sub-toxic levels that reduce synaptic density or disrupt neuritic network organisation.
Excitotoxicity is a key pathophysiological mechanism within the ischemic cascade following stroke. It emerges within minutes to hours of stroke onset and persists throughout post‑ischemic phases (inflammation, synaptic remodelling, and epileptogenic hyperexcitability).
Glutamate-induced toxicity: organ-on-chip
Excitotoxicity is a key pathophysiological mechanism in ischemic stroke and contributes significantly to the consolidation of brain injury at sites distant from the ischemic core.
Our brain‑on‑chip model is designed to reproduce secondary excitotoxicity by delivering glutamate specifically into a synaptic chamber located at the interface between two spatially separated neuronal populations (i.e. each maintained in its own microphysiological environments).
Our microfluidic platform, combined with high‑content imaging, enables medium‑throughput screening of the effects of your compound on an isolated synapse, physically distant from the soma and exposed to excessive glutamate.
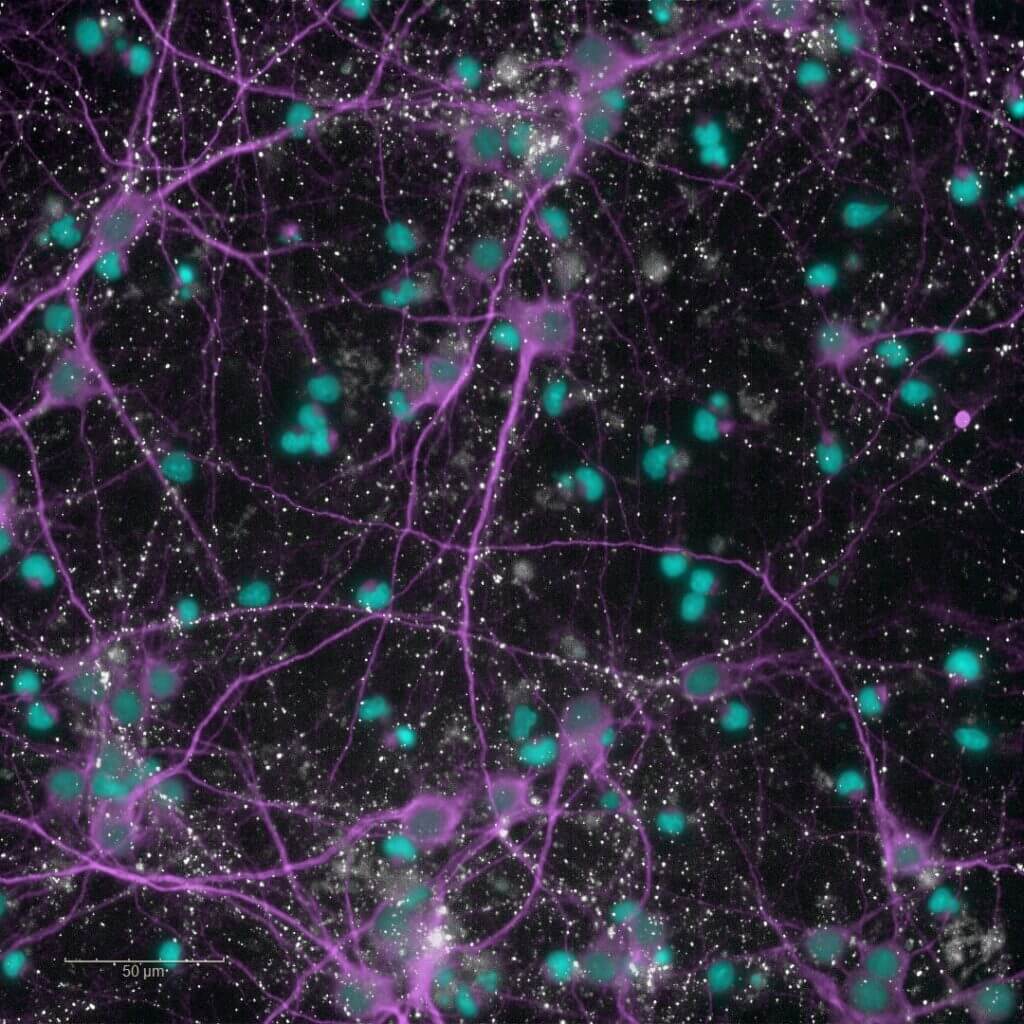
Glutamate uptake by astrocytes
Astrocytes play a central role in maintaining synaptic homeostasis and preventing excitotoxicity by clearing excess extracellular glutamate. This protective function becomes impaired in reactive astrocytes during the post-ischemic inflammatory phase.
Our astrogliosis model enables the screening of your compounds targeting glutamate uptake in human astrocytes (iPSC) activated with a cytokines cocktail. Activation of the NFκB pathway is also available as a routine readout through our high-content imaging platform.
Pharmacological interaction and hemostasis
Clot lysis assay
Documenting potential interactions between reference fibrinolytics and drug candidates is required by regulatory agencies when these treatments are intended for combined administration during the acute phase of stroke.
Using turbidimetry in 96-well plates, the clot lysis assay quantifies fibrinolytic activity in human plasma pools, relying on parameters such as the 50% clot lysis time (LT50). The high reproducibility of the assay ensures reliable and straightforward comparison of dose-response curves for Alteplase or Tenecteplase, either alone or in combination with multiple concentrations of your compound.
ROTEM assay
This assay also allows investigation of compound interactions when combined with reference fibrinolytics (Alteplase, Tenecteplase).
The ROTEM assay, performed on fresh human whole blood, measures the viscoelastic properties of the clot throughout its formation and subsequent lysis.
A wide range of parameters can be extracted from the thromboelastogram, including the clotting time, the maximum clot firmness, and the 50% clot lysis time (LT50). This low-throughput assay offers an extensive and customisable set of hemostatic conditions for evaluating anticoagulant or thrombolytic compounds, including extrinsic or intrinsic activation pathways, as well as the addition of coagulation factors or modulators.
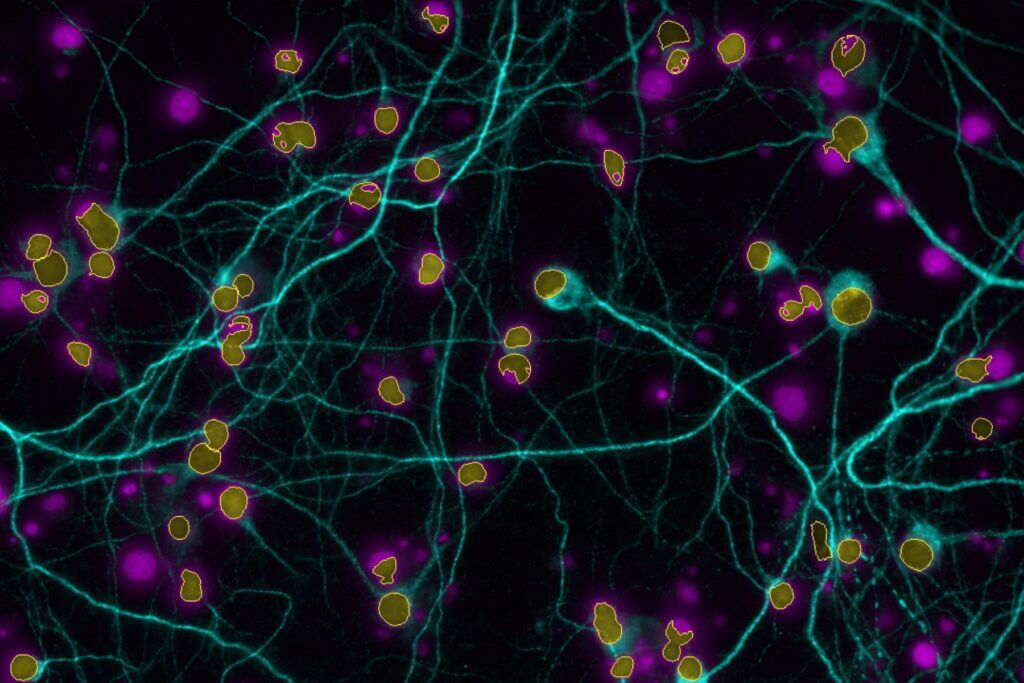
High-Content Imaging
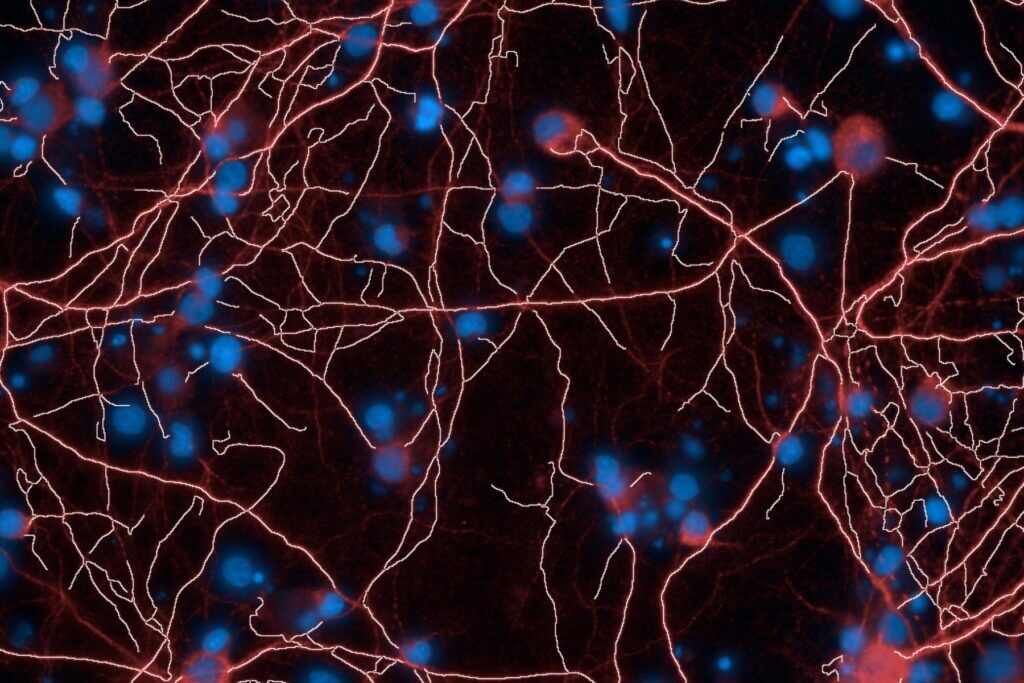
Brain‑on‑Chip platform

Small and Large Animal Facilities
Preclinical Medical Imaging

Immunohistochemistry, Immunofluorescence, and Histology
Immunoassays
The effects of your molecules can be evaluated through complementary and multi-scale approaches.