in vivo pharmacology models in Cardiovascular diseases
For more than 25 years, SYNCROSOME has been offering support in conducting in vivo pharmacology studies targeting cardiovascular and cardiometabolic diseases. We offer a comprehensive portfolio of rodent models of heart failure and pulmonary arterial hypertension (PAH). In addition, we have several models of heart failure with preserved ejection fraction (HFpEF), each associated with specific features of metabolic syndrome—including obesity, diabetes, hypertension, and kidney disease—alongside fibrosis and inflammation.
We provide you with our expertise, technical capabilities, and dedicated platforms for small‑animal surgery and imaging to support the evaluation of your lead compounds and drug candidates.
Our models are validated with a panel of reference compounds and are suitable for evaluating both symptomatic treatments and disease‑modifying therapies.
Right heart failure
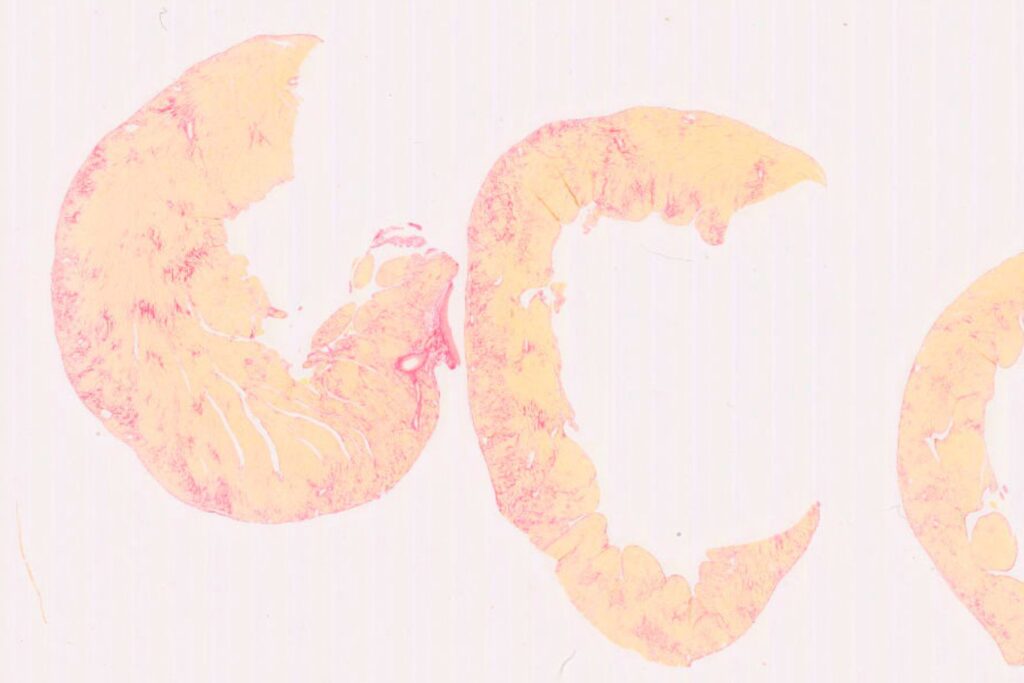
Right heart failure is characterized by the inability of the right ventricle to effectively pump blood toward the lungs. This dysfunction often results from a chronic increase in pressure within the pulmonary artery, as seen in pulmonary arterial hypertension (PAH). Faced with this persistent overload, the right ventricle undergoes remodeling, progressively leading to impaired cardiac function.
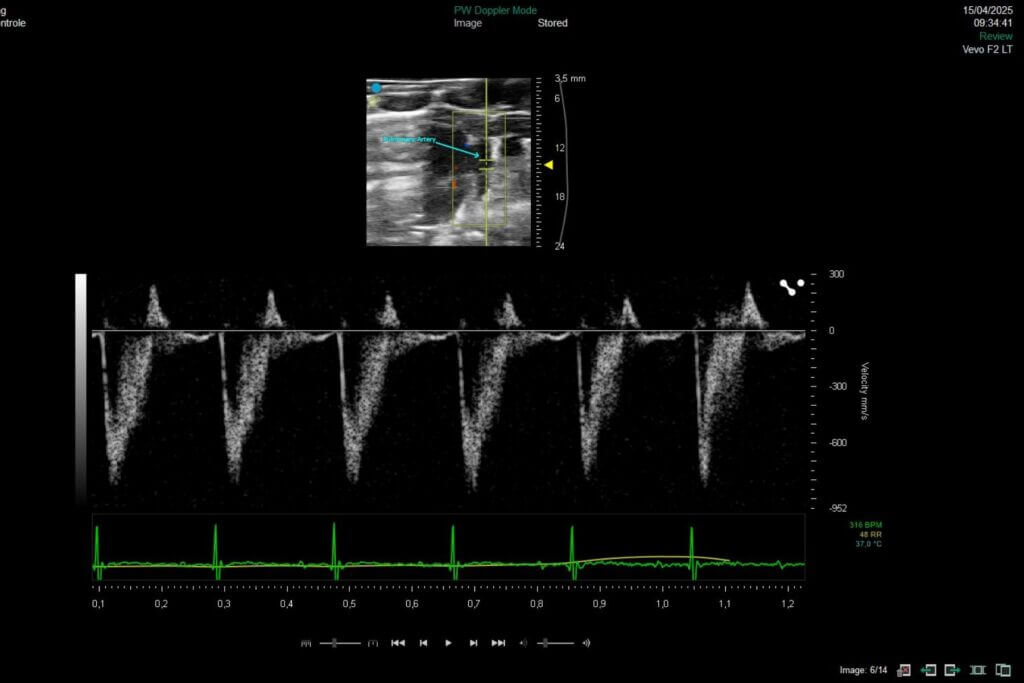
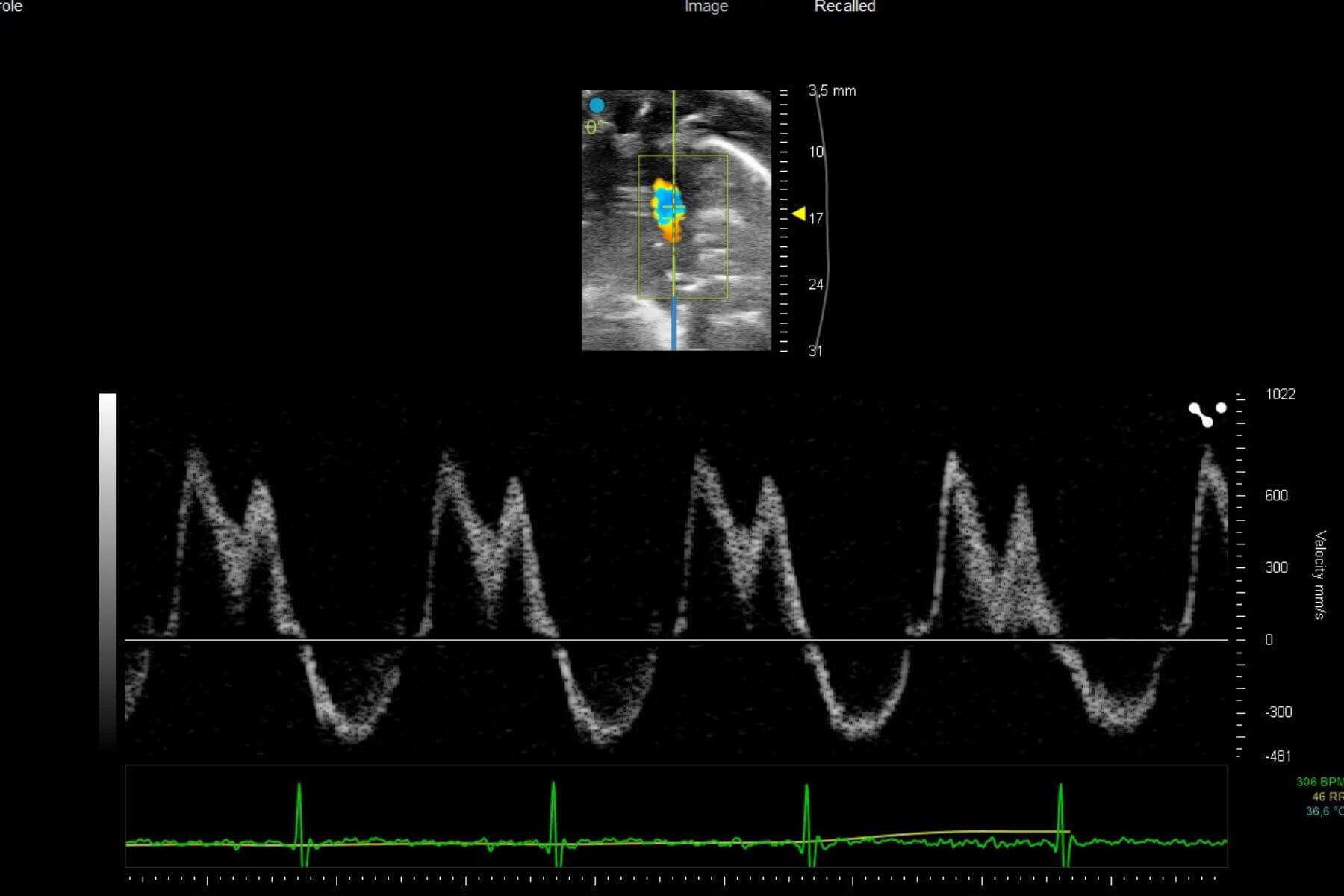
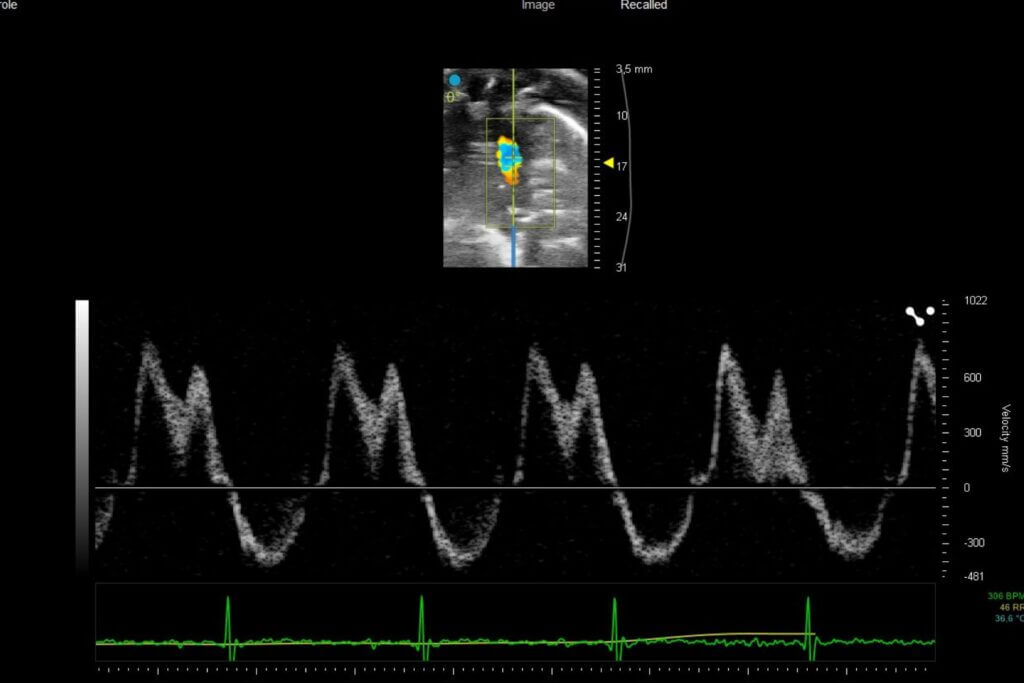
Syncrosome’s portfolio enables you to evaluate the effects of your drug candidates in recognized and validated pathological models designed for the pharmacological assessment of cardiac function, as well as cardiac and pulmonary arterial tissue remodeling.
Pulmonary arterial hypertension (PAH)
Hypoxia model
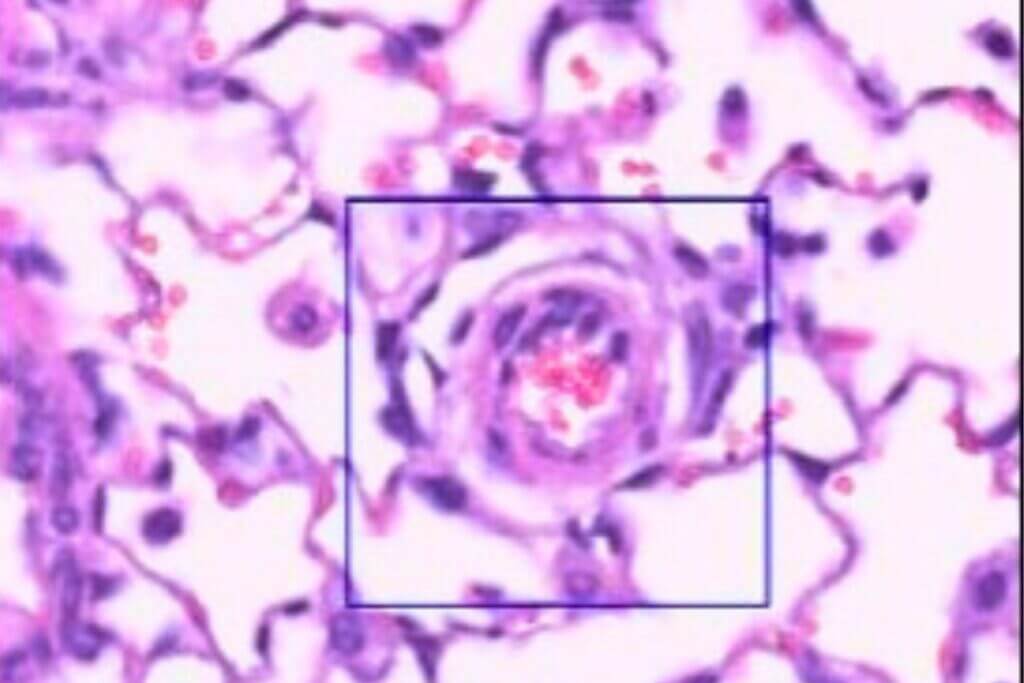
The hypoxia model is a classic approach to induce moderate pulmonary arterial hypertension in animals. It relies on the induction of chronic hypoxia using specially designed hypoxia chambers. The model is characterized by persistent pulmonary vasoconstriction, followed by moderate remodeling of the pulmonary arteries.
Rat model.
Sugen-Hypoxia
The SUGEN‑hypoxia model combines the injection of a VEGF receptor inhibitor with prolonged exposure to hypoxia to induce progressive and severe pulmonary hypertension accompanied by right‑ventricular heart failure. This model reproduces most of the clinical features observed in humans, including plexiform lesions in the pulmonary arterioles. It is considered the reference model for studying advanced forms of PAH, allowing the evaluation of therapies targeting complex vascular remodeling, cell proliferation, or endothelial dysfunction. Its severity and stability make it a powerful tool for generating highly translational preclinical data.
Rat model.
Monocrotaline (MCT)
The monocrotaline (MCT) model is one of the most widely used approaches for the experimental induction of PAH. A single injection of MCT causes marked endothelial and inflammatory injuries in the pulmonary arterioles. Mimicking the human disease, the model is characterized by progressive vascular remodeling and right‑ventricular hypertrophy. This quick model is useful for the rapid screening of therapies targeting various key features of PAH. Its well‑characterized kinetic allows optimization of therapeutic windows and the generation of robust data to demonstrate the efficacy of your drug candidates.
Rat model.
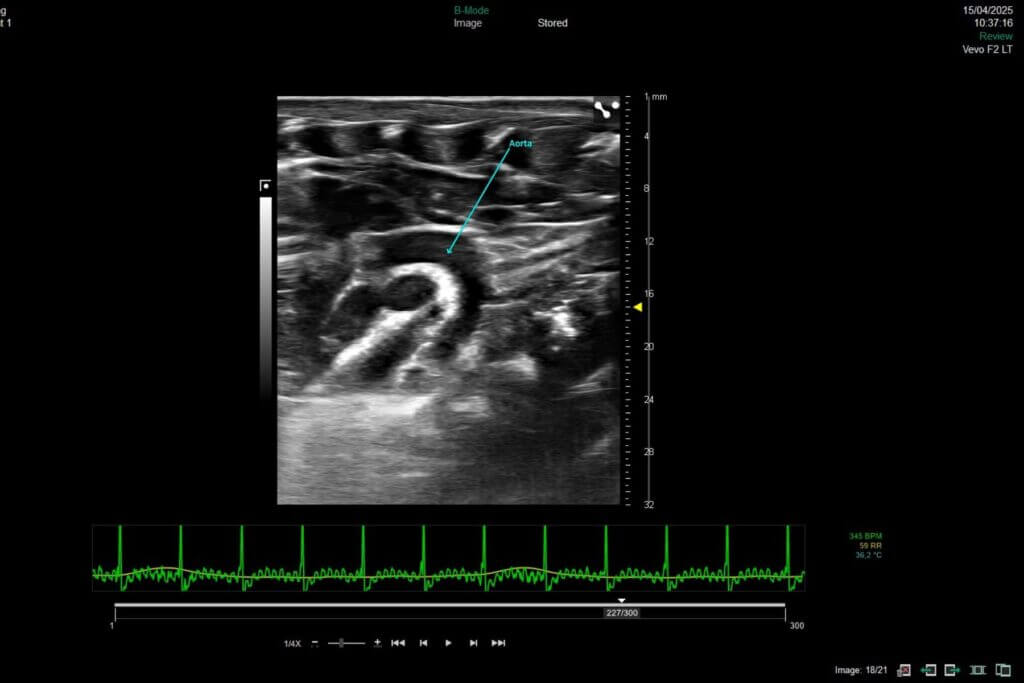
Pulmonary embolism
Pulmonary embolism model induced by injection of autologous clots
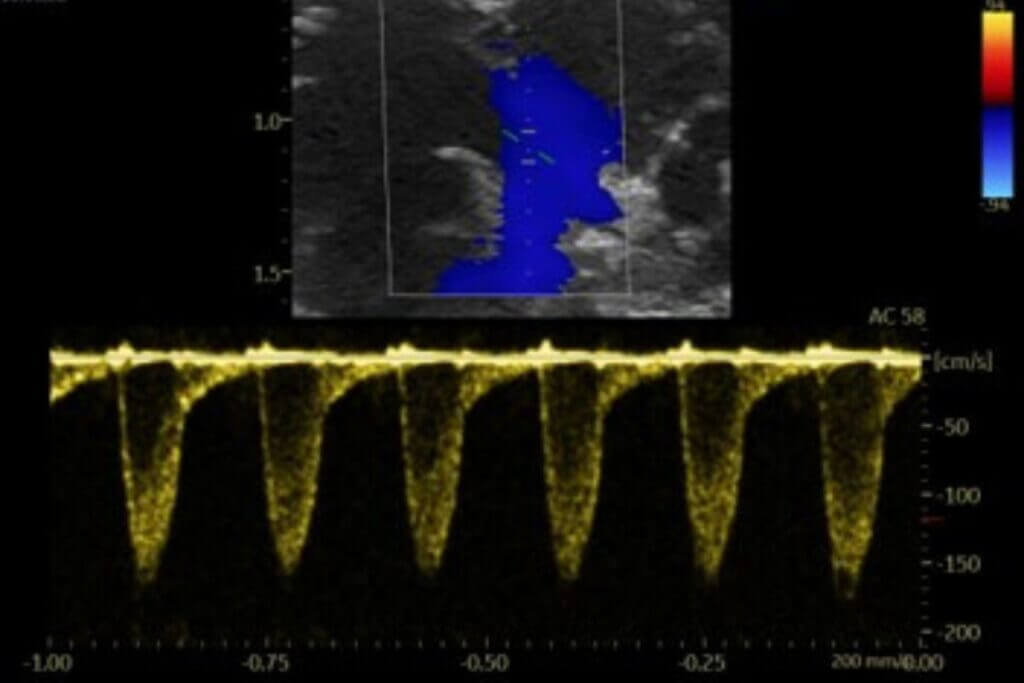
The pulmonary embolism model induced by injection of autologous clots relies on preparing and injecting pre‑formed calibrated clots into the jugular vein to selectively obstruct the pulmonary arteries. This model reproduces the acute increase in pulmonary pressure observed in human clinical settings, along with significant stress on the right ventricle and secondary vascular remodeling. The severity of the condition can be adjusted based on the amount of thrombotic material used. Its strong reproducibility and rapid implementation make it suitable for quickly evaluating the efficacy of thrombolytic, vasodilatory, or cardioprotective treatments.
Rat model.
Left heart failure
Left heart failure is characterized by an impaired ability of the heart to deliver sufficient cardiac output to meet the demands of the systemic circulation. It results from chronic hemodynamic stress, such as increased afterload, or ischemic myocardial injury, and is associated with progressive structural and functional cardiac remodeling.
Our platform enables the direct evaluation of therapeutic candidates in both HFrEF and HFpEF models, integrating comprehensive assessments of left ventricular function by echocardiography, cardiac remodeling, and cardio-renal-metabolic interactions.
Left heart failure with reduced ejection fraction (HFrEF)
In many patients with extensive ischemic heart disease or myocardial infarction, the pathology progresses toward a reduction in left ventricular ejection fraction defining HFrEF.
Our rat models reproduce key characteristics of the human HFrEF.
Permanent coronary artery ligation model
The permanent left coronary artery ligation model is based on the definitive ligation of this artery, inducing a large myocardial infarction and progressive left‑ventricular failure. It recapitulates key mechanisms of post‑infarction remodeling: necrosis, inflammation, fibrosis, and long‑lasting impairment of cardiac function. This model is a robust tool for evaluating cardioprotective, antifibrotic, or regenerative therapies.
Rat model.
Ischemia-reperfusion injury model (IRI)
The myocardial ischemia–reperfusion (IRI) model consists in transient occlusion of the left coronary artery to reproduce the damage caused by tissue reperfusion following a treated myocardial infarction. This approach generates massive oxidative stress, acute inflammation, significant apoptosis, and early myocardial remodeling. This model provides an ideal platform for evaluating cardioprotective, antioxidant, or anti‑inflammatory therapies targeting reperfusion‑induced injury.
Rat model.
Isoproterenol-induced cardiac injury model
The isoproterenol‑induced cardiac injury model is based on the repeated administration of this synthetic β‑agonist, which triggers acute cardiac stress leading to myocardial necrosis, marked inflammation, and rapid left‑ventricular remodeling. It reproduces diffuse lesions similar to those observed in myocarditis or stress‑induced cardiomyopathies. This quick model make it an effective tool for a rapid screening of cardioprotective, anti‑inflammatory, or antifibrotic therapies.
Rat model
Left heart failure with preserved ejection fraction (HFpEF)
HFpEF is driven by impaired ventricular filling due to increased myocardial stiffness, despite preserved systolic function. HFpEF is closely associated with metabolic comorbidities, including obesity, type 2 diabetes, and metabolic syndrome, as well as with chronic kidney disease, which contributes to disease progression.
Our rodent models recapitulate key HFpEF-associated cardiac dysfunction across multiple combinations of metabolic comorbidities.
Dahl rat model
The salt‑sensitive Dahl rat model progressively and physiologically reproduces the mechanisms of HFpEF. When exposed to a high‑salt diet, these animals develop severe hypertension, concentric left‑ventricular remodeling, myocardial fibrosis, and impaired cardiac relaxation, while maintaining a preserved ejection fraction.
Rat model.
BTBR ob/ob mouse
The BTBR ob/ob mouse model reproduces a metabolic form of HFpEF, characterized by severe obesity, insulin resistance, renal dysfunction, and progressive diastolic impairment. These animals develop concentric left‑ventricular remodeling, myocardial fibrosis, and impaired cardiac relaxation, while maintaining a preserved ejection fraction.
Mouse model.
db/db mouse
The db/db mouse model reproduces a metabolic form of HFpEF associated with severe obesity and type 2 diabetes. These animals develop insulin resistance, systemic inflammation, myocardial fibrosis, and progressive impairment of ventricular relaxation, while maintaining a preserved ejection fraction.
Mouse model.
L-NAME + high fat diet model
The combined L‑NAME and high‑fat diet (HFD) model reproduces a multifactorial form of HFpEF, combining hypertension, endothelial dysfunction, obesity, and systemic inflammation. Inhibition of NO synthesis by L‑NAME induces a marked increase in blood pressure, while the HFD promotes the development of a metabolic syndrome with concentric remodeling, myocardial fibrosis, and diastolic dysfunction, all while maintaining a preserved ejection fraction.
Mouse model.
Fibrinolysis and anticoagulants
Pulmonary embolism
Pulmonary embolism is defined as the sudden obstruction of one or more pulmonary arteries, most often by a blood clot. This blockage reduces blood flow to part of the lung, leading to impaired gas exchange and acute overload of the right ventricle.
Pulmonary embolism model induced by injection of autologous clots
The pulmonary embolism model induced by injection of autologous clots relies on preparing and injecting pre‑formed calibrated clots into the jugular vein to selectively obstruct the pulmonary arteries. This model reproduces the acute increase in pulmonary pressure observed in human clinical settings, along with significant stress on the right ventricle and secondary vascular remodeling. The severity of the condition can be adjusted based on the amount of thrombotic material used. Its strong reproducibility and rapid implementation make it suitable for quickly evaluating the efficacy of thrombolytic, vasodilatory, or cardioprotective treatments.
Rat model.
Thromboembolic stroke
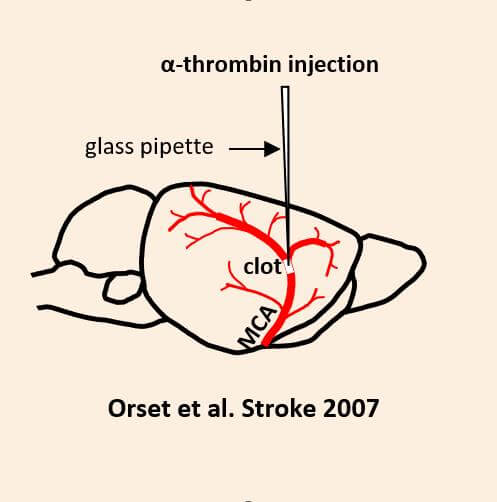
MCA occlusion by thrombin injection
This preclinical ischemic stroke model is induced by the formation of a fibrinrich clot in the MCA. This clot is efficiently lysed by standard clinical fibrinolytics (Alteplase, Tenecteplase), reflecting the patient subgroup that responds to standard-of-care fibrinolytics. This makes the model a key translational tool for assessing the efficacy of your thrombolytic candidates. This model is also highly relevant for evaluating neuroprotective compounds in an experimental setting that closely mimics the clinical reality of the acute phase of stroke (with the presence of a thrombus and pharmacological recanalization achieved within a clinically relevant treatment window). It additionally enables the study of interactions with reference fibrinolytic treatments.
Validated model available in mice.
MCA occlusion by FeCl₃ or AlCl₃ application
This preclinical ischemic stroke model is induced by the formation of a platelet‑rich clot that is resistant to standard fibrinolytics (Alteplase, Tenecteplase) but responsive to other thrombolytic agents such as N‑acetylcysteine. By reflecting the subpopulation of patients who fail to respond to standard‑of‑care treatments, this translational model is a powerful tool for assessing the efficacy and superiority of your candidates, combination therapies, and pharmacological interactions.
Validated model available in mice.
tMCAO model by autologous clot injection
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
ICH induced by collagenase injectionStereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Validated model available in rats.
Hemorrhagic stroke
ICH induced by collagenase injection
Stereotaxic injection of collagenase rapidly disrupts the vascular endothelium, inducing a reproducible hemorrhage with the formation of an intracerebral hematoma, accompanied by an inflammatory response and clear neurological deficits. This ICH model provides a valuable translational tool for assessing the efficacy of your drug candidates on bleeding, neuroinflammation, and neuroprotection. Depending on your needs, it also offers the possibility to evaluate the risk of rebleeding or the effectiveness of antidotes across a panel of reference anticoagulants and fibrinolytics.
Bleeding model
Tail bleeding assay
The tail bleeding test is a goldstandard assay used to evaluate hemostasis and platelet function in vivo.
This model is sensitive to anticoagulant and fibrinolytic treatments commonly administered in the clinic, such as heparin, direct oral anticoagulants, or Alteplase. This model supports the evaluation of candidate efficacy across multiple drug classes, such as anticoagulants, procoagulants, and antiplatelet agents.
Validated model available in mice
Pharmacokinetics (PK) and neuro-PK
Our pharmacokinetic studies are based on flexible kinetic models tailored to your drug candidate. We analyze standard biological matrices (plasma, serum, CSF) as well as key organs or anatomical regions to accurately characterize your molecule’s tissue exposure. These robust and customizable approaches provide a solid foundation for your preclinical decisions.
PK model
We offer studies tailored to your PK and Neuro-PK needs, covering various administration routes (subcutaneous, intraperitoneal, intravenous, oral, intranasal, etc.) in both acute and chronic treatment paradigms. The bioavailability of your molecules is quantified across different compartments—including blood, brain, cerebrospinal fluid, or any other tissue of interest. This approach provides reliable data to support decision-making and accelerate the development of your therapeutic candidates.
Available in mice, rats, and pigs.
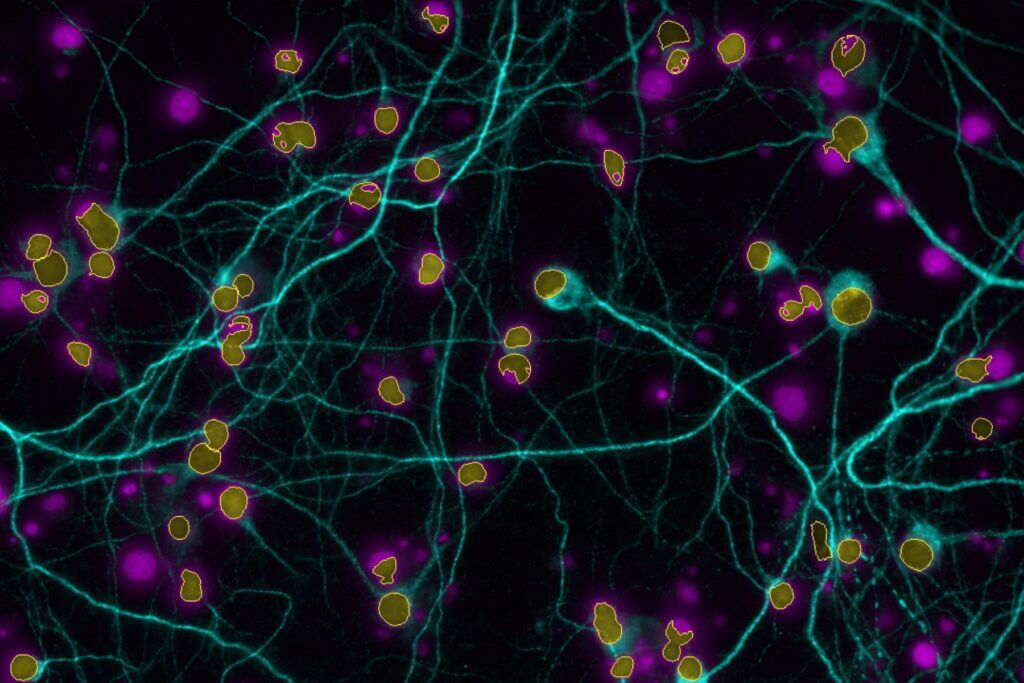
High-Content Imaging
Brain‑on‑Chip platform

Small and Large Animal Facilities
Preclinical Medical Imaging

Immunohistochemistry, Immunofluorescence, and Histology
Immunoassays
The effects of your molecules can be evaluated through complementary and multi-scale approaches.