in vivo pharmacology models in Neurology & the CNS
For more than 35 years, BRAINXPLORE has been providing robust and reproducible preclinical neurology services, supported by a comprehensive portfolio of gold‑standard in vivo models to evaluate the efficacy of your lead compounds and drug candidates in CNS disorders. We assess the effects of your molecules using complementary, multi‑scale approaches ranging from molecular biology to behavior.
Every element of the study design — route of administration, timing, dosing regimen, readouts, group size, and inclusion of reference compounds — can be fully customized to meet your specific needs.
Neurodegenerative diseases
We provide a portfolio of mouse models designed to accelerate drug efficacy testing for Alzheimer’s disease (AD) and Parkinson’s disease (PD). Our proprietary human oligomers allow you to evaluate the ability of your therapeutic candidates to modulate proteinopathies and improve translational success.
Alzheimer’s disease
Neurodegeneration induced by amyloid-β(1-42) oligomers combined with aging
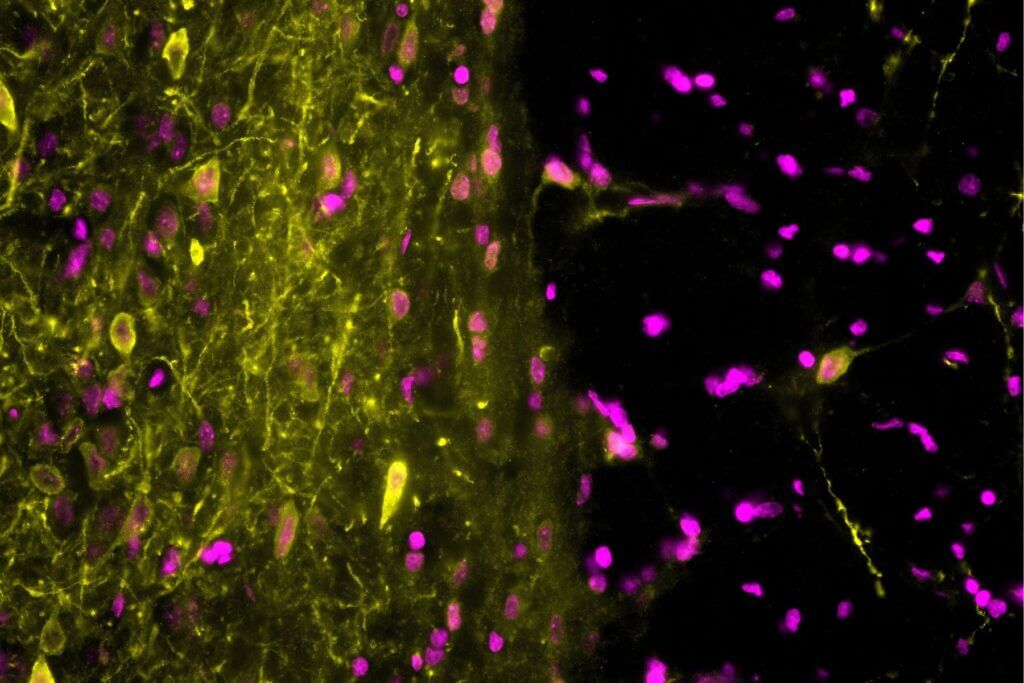
In aged mice, intracerebral injection of amyloid-β(1-42) oligomers induces robust short‑ and long‑term memory impairments, pronounced neurodegeneration, and marked neuroinflammation across the hippocampus and cortex, closely reproducing key features of the Alzheimer‑like phenotype. By positioning aging as a key driver of disease progression, this model provides strong predictive value for clinical translation and offers a powerful, industry-ready platform to assess the therapeutic potential of your candidates under conditions closely aligned with human pathophysiology.
Neurodegeneration induced by amyloid-β(1-42) oligomers
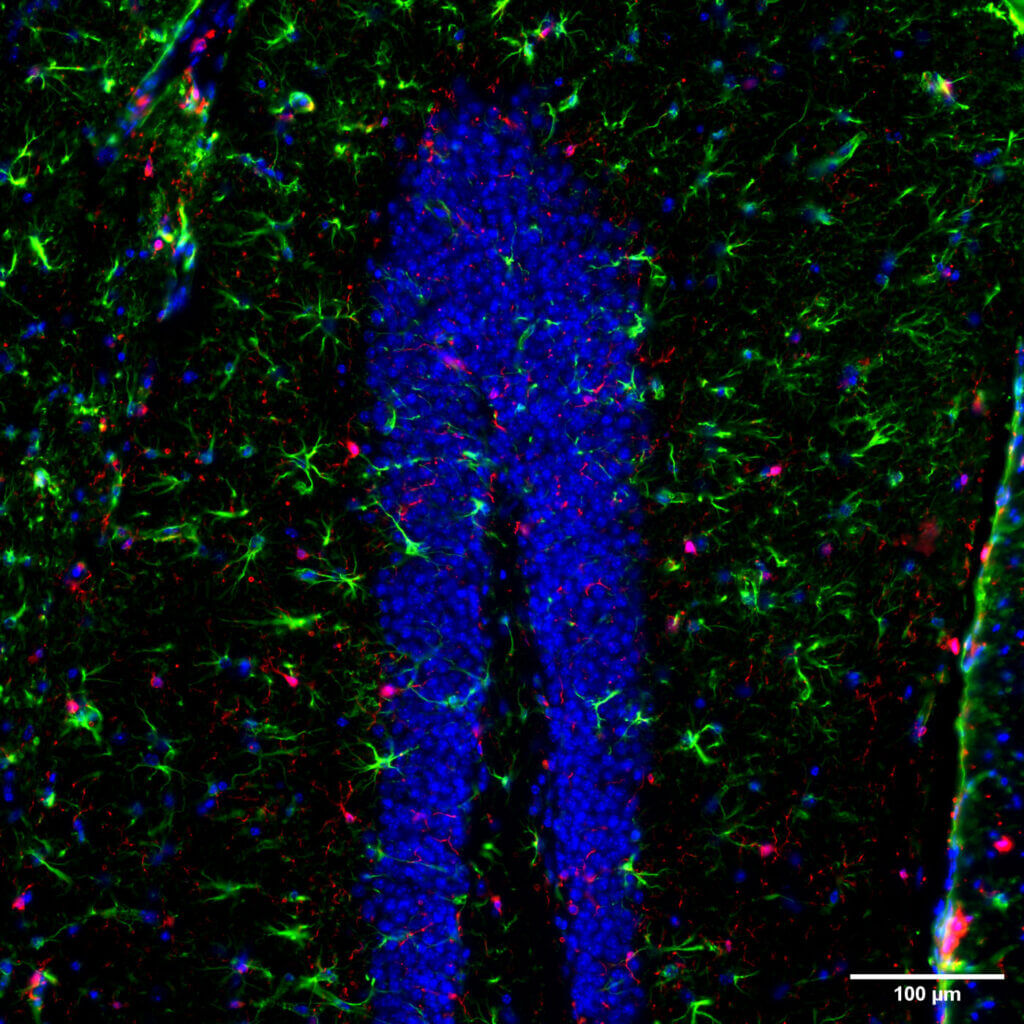
Intracerebral injection of amyloid-β(1-42) oligomers in mice induces synaptic loss, neuroinflammation, and neurodegeneration in the hippocampus and cortex, reproducing the key features of Alzheimer’s disease. This model provides a fast and reliable approach to investigate the pathological mechanisms associated with amyloid-β(1-42) oligomers and to support early‑stage screening of your therapeutic candidates.
Scopolamine-induced amnesia
Administration of scopolamine, a muscarinic receptor antagonist, induces episodic and spatial memory deficits in mice, mimicking key features of cognitive impairments. This model provides a fast and reliable approach to demonstrate the efficacy of your candidates targeting memory enhancement.
Parkinson’s disease
Dopaminergic neuron degeneration induced by intranigral injection of the neurotoxin 6-OHDA
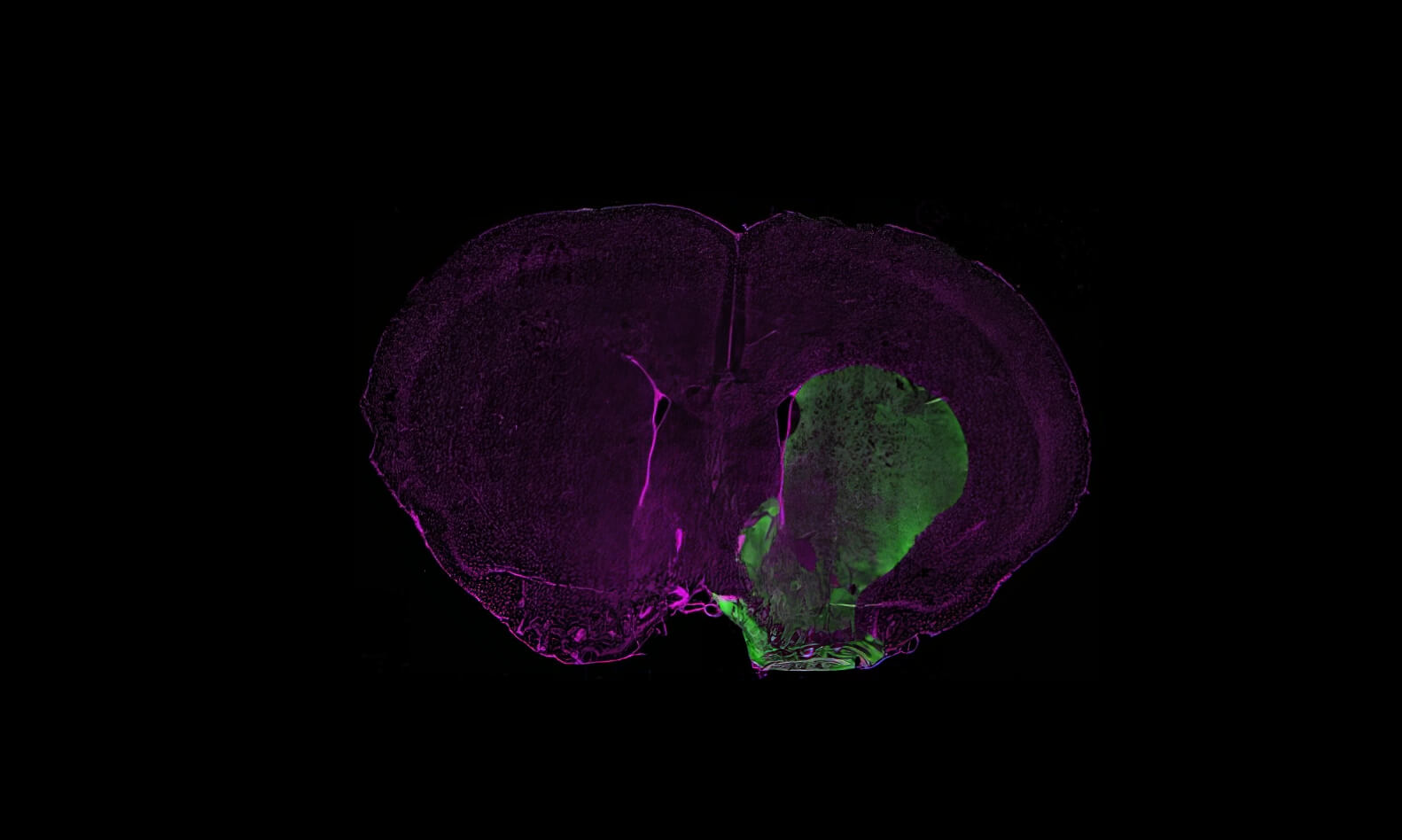
The unilateral injection of 6‑OHDA specifically targets the SNpc, leading to an almost complete lesion of the nigrostriatal pathway. The lesioned animals exhibit motor impairments, including akinesia, a selective loss of dopaminergic neurons, and neuroinflammation.
Dopaminergic neuron degeneration induced by intranigral injection of the neurotoxin MPP⁺
The unilateral injection of MPP⁺ specifically targets the SNpc, leading to an almost complete lesion of the nigrostriatal pathway. The lesioned animals exhibit motor impairments, including akinesia, a selective loss of dopaminergic neurons, and neuroinflammation.
Behavioral pharmacology
Our extensive portfolio of behavioral tests covers a broad range of CNS‑related functions, including sensorimotor skills, cognition, mood, pain, itch, well‑being, and motivation‑oriented behaviors. Through our dedicated platform, we provide highly reproducible behavioral measurements to quantify performance and infer brain functions. Most of our tests can be used as stand‑alone assessments or integrated as readouts within our disease models.
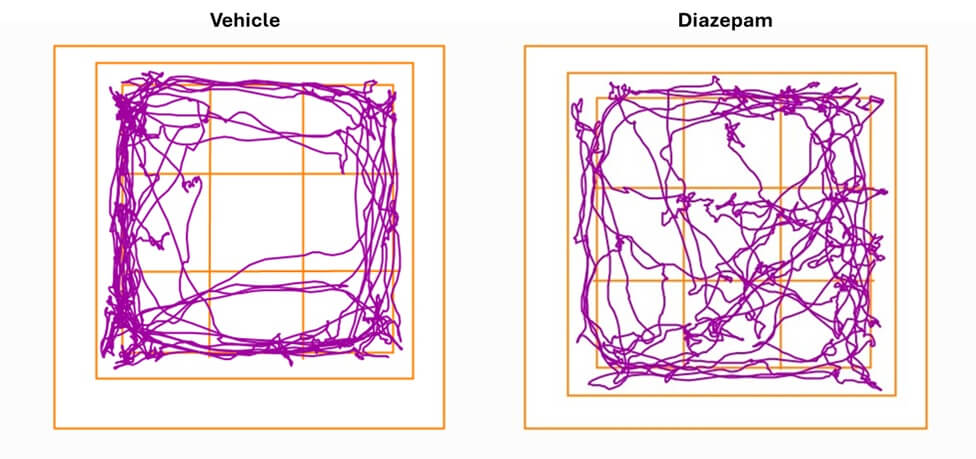
Anxiety
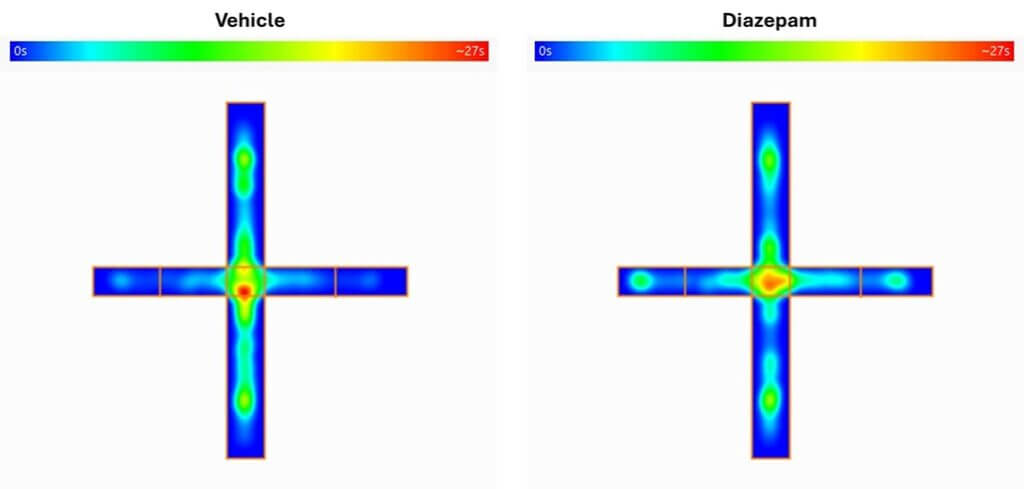
Elevated plus maze test
Based on a conflict between the innate tendency to explore new environments and the aversion to open spaces, the Elevated Plus Maze test is a gold-standard for assessing anxiety-like behavior in rodents. This test enables the screening of anxiolytic compounds and can be combined with pathology models to evaluate state anxiety.
Validated in rats.
Light/Dark box test
Based on a conflict between the innate tendency to explore novel environments and the aversion to light, the light–dark box test is a gold-standard assay used to measure anxiety-like behavior in rodents. This test enables the screening of your anxiolytic compounds and can be combined with pathology models to evaluate state anxiety.
Validated in mice.
Conditioned defensive burying test
The conditioned defensive burying test relies on the natural behavior of rodents which consists of projecting bedding material toward a potentially aversive stimulus. In laboratory conditions, introducing a novel and aversive object into a familiar environment triggers this burying behavior in rats. This behavior is directly related to the animals’ level of anxiety, making it a reliable assay for screening anxiolytic compounds.
Validated in rats.
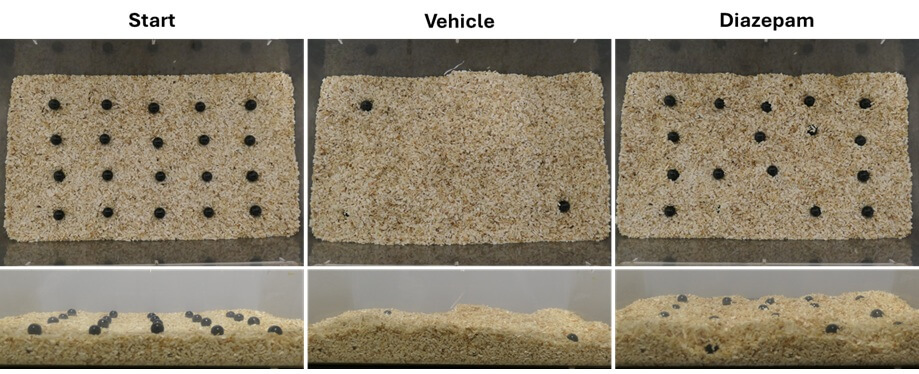
Marble burying test
The marble burying test relies on the natural tendency of mice to bury unfamiliar objects introduced into a familiar environment. This behavior correlates with the animals’ anxiety level and is widely used to screen anxiolytic compounds.
Validated in mice.
Depression
Sucrose preference test (anhedonia)
The sucrose preference test relies on the innate tendency of rodents to prefer sweetened water over plain water. This behavioral paradigm is widely interpreted as a measure of anhedonia in animals, as the natural preference for sucrose diminishes under chronic stress or severe pathology and can be restored by treatment with reference antidepressant compounds.
For the evaluation of your antidepressant candidates, anhedonia is induced through chronic stress exposure. This test can also be integrated into other pathology models (such as stroke) to measure depressive-like behavior in animals within a defined pathological context.
Validated in rats.
Well-being
Cognition
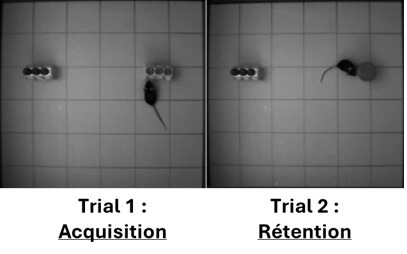
Novel object recognition
The object recognition test allows the assessment of working memory and the detection of mnemonic deficits in mice. This paradigm, based on the innate tendency of rodents to preferentially explore a novel object rather than a familiar one, is useful for evaluating the efficacy of your candidates on cognition.
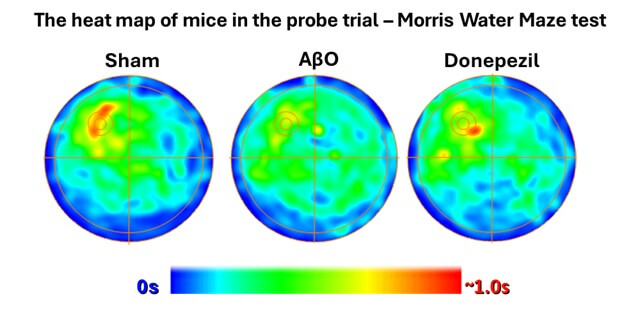
Morris water maze
The Morris water maze is widely used to evaluate spatial reference memory in rodents and to predict the efficacy of your therapeutic candidates on cognitive performance. This paradigm, based on the animal’s motivation to escape the aversive nature of water, provides data on its ability to navigate and locate the escape platform. As these performances are hippocampus-dependent, this test is particularly suited for the study of cognitive impairments in Alzheimer’s disease models.
Validated in mice and rats.
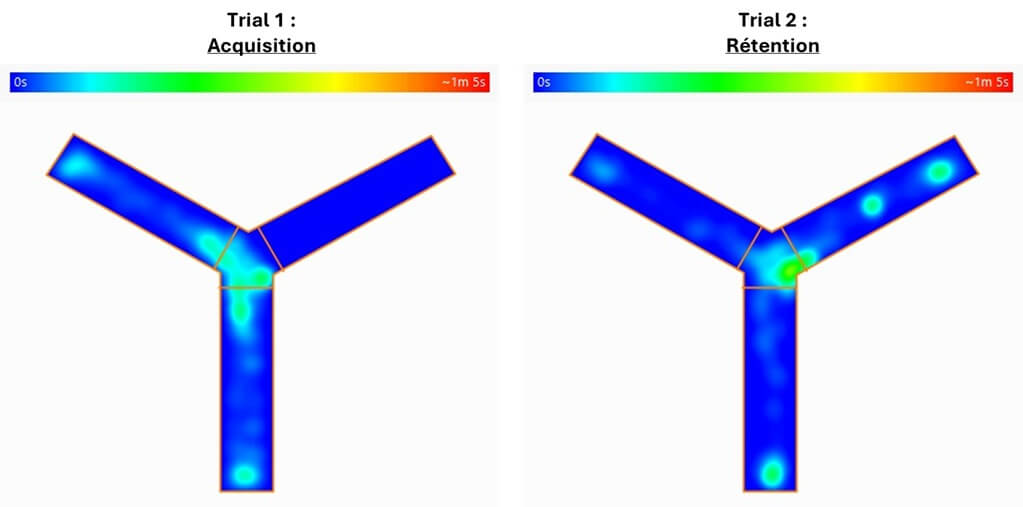
Y-maze
The Y-maze is a rapid method for measuring spatial reference memory and detecting mnemonic deficits in rodents. This test, which leverages the spontaneous tendency of rodents to explore a novel environment rather than a familiar one, is useful for evaluating the efficacy of therapeutic candidates on cognition during the early stages of your preclinical studies.
Itching and Pain
Itching test
The sensation of itch is a common feature of many dermatological and CNS disorders (neuropathic itch). BrainXplore has developed strong expertise in the behavioral assessment of itch in rodents. Depending on the mechanism of action of your compound, itch can be induced through various methods.
Pain upon skin application
Following skin injury, cutaneous pain can result from direct nociceptor activation or inflammation that activate nociceptive pathways. This model evaluates acute tolerance to topical formulation application under physiologically relevant conditions.
Tail-flick test
The tail‑flick test is a rapid and ethically acceptable method for assessing acute thermal antinociception in rodents. It is widely used to screen the analgesic efficacy of drug candidates. Morphine is proposed as a reference compound.
Sensorimotor
Corridor test
The Corridor test is used to assess tactile sensory asymmetry resulting from a lateralized lesion of the sensorimotor cortex. The test relies on the exploration of multiple series of objects placed along the walls of a corridor using vibrissae: a reduction in contralesional sensitivity is reflected by fewer exploratory responses on the affected side. The Corridor test is a relevant tool for monitoring sensorimotor recovery and quantifying the effects of your drug candidates on post‑lesional impairments, particularly after stroke.
Ladder test
The Ladder test is used to assess fine motor coordination and limb‑placement accuracy in rodents. As the animal progresses freely across a ladder with irregularly spaced rungs, it must continuously adjust the position of its paws. Because the test is highly sensitive to central nervous system impairments, quantifying foot‑placement errors provides a reliable evaluation of locomotor deficits and limb‑use asymmetry following brain injury. This assay offers a robust readout for assessing the effects of your drug candidates on functional recovery.
Cylinder test
The cylinder test is used to assess the sensorimotor asymmetry induced by a unilateral lesion of the nigrostriatal pathway. This test relies on the animal’s natural exploratory behavior when placed in a novel environment. The spontaneous use of each forelimb is quantified when the rat is introduced into a transparent cylinder, which makes it possible to effectively detect motor asymmetry induced by the lesion and serves as a relevant tool for evaluating the therapeutic effects of pharmacological compounds or neuroprotective interventions. Moreover, it is considered one of the gold‑standard behavioral tests in the rat 6‑OHDA model.
Open field test
The open field test assesses locomotor activity in rodents under conditions of spontaneous exploration. It is commonly combined with other behavioral assays to control basal locomotor activity or included as a readout in disease models to evaluate locomotor performance and motivation to explore. As a stand‑alone test, it can be used to evaluate the locomotor effects of your psychoactive drug.
Rotarod test
The Rotarod test is used to evaluate motor coordination and balance in rodents during a forced‑walking task on a rotating drum. It is primarily employed in our Parkinson’s disease and stroke models to measure motor performance. This test is a relevant tool for quantifying the effects of your drug candidates on functional recovery and fatigability.
Grip test
The grip test provides a robust quantification of forelimb grip strength in rodents. It is used to assess muscle strength deficits across various conditions, including central nervous system (CNS) lesions such as post‑stroke hemiparesis. This assay is a sensitive and relevant endpoint for evaluating the effects of your drug candidates on the recovery of muscle strength deficits.
Neuroscore
Specially developed and validated by our team, our neuroscore provides an integrated tool for assessing sensorimotor performance in rats. Based on a structured neurological examination comprising 15 items (responses to visual, auditory and tactile stimuli, locomotor evaluation, etc.), it enables a fine and reproducible quantification of neurological deficits resulting from central nervous system lesions. Designed in line with clinical reference scales such as the NIHSS, this neuroscore strengthens the translational relevance of our stroke models. It offers a sensitive readout to measure the effects of your drug candidates on functional recovery.
Stepping test (or adjusted step test)
The Stepping test detects deficits in forelimb motor initiation resulting from CNS impairment, particularly damage to the nigrostriatal pathway. This test reliably reflects limb akinesia and locomotor disturbances characteristic of Parkinson’s disease during a voluntary movement. The Stepping test is also highly sensitive to L‑DOPA, the gold‑standard treatment in Parkinsonian patients. It is therefore a relevant tool for evaluating the therapeutic efficacy of new compounds targeting motor deficits associated with dopaminergic degeneration.
Stroke
Our stroke models—hemorrhagic (IHC), distal ischemic, and proximal ischemic (MCAO)—are highly relevant for your POC, PK/PD, superiority efficacy studies, combination therapy studies, pharmacological interaction assessments, and assessment of hemorrhagic transformation risk.
Trust our validated and well-established animal models to accelerate the selection of your drug candidates with STROK@LLIANCE services.
Stroke models
Find our ischemic and hemorrhagic stroke animal models for testing your drug candidates on our page dedicated to our preclinical services in neurovascular diseases.
Pharmacokinetics (PK) and neuro-PK
Our pharmacokinetic studies are based on flexible kinetic models tailored to your drug candidate. We analyze standard biological matrices (plasma, serum, CSF) as well as key organs or anatomical regions to accurately characterize your molecule’s tissue exposure. These robust and customizable approaches provide a solid foundation for your preclinical decisions.
PK model
We offer studies tailored to your PK and Neuro-PK needs, covering various administration routes (subcutaneous, intraperitoneal, intravenous, oral, intranasal, etc.) in both acute and chronic treatment paradigms. The bioavailability of your molecules is quantified across different compartments—including blood, brain, cerebrospinal fluid, or any other tissue of interest. This approach provides reliable data to support decision-making and accelerate the development of your therapeutic candidates.
Available in mice, rats, and pigs.
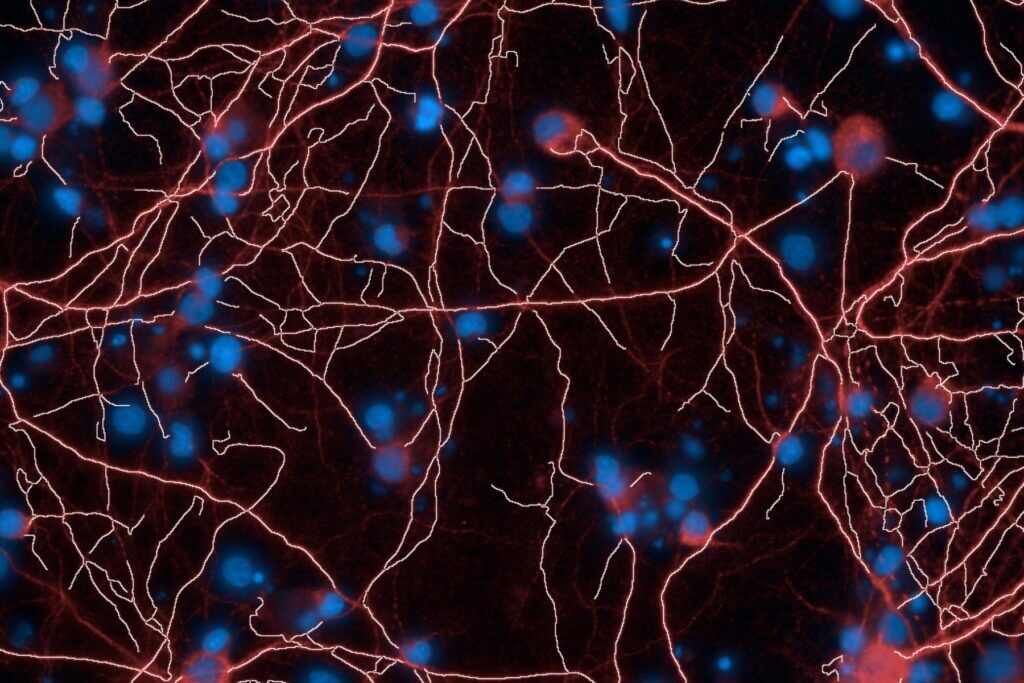
BRAINXPLORE integrates cutting-edge technologies for translational research.
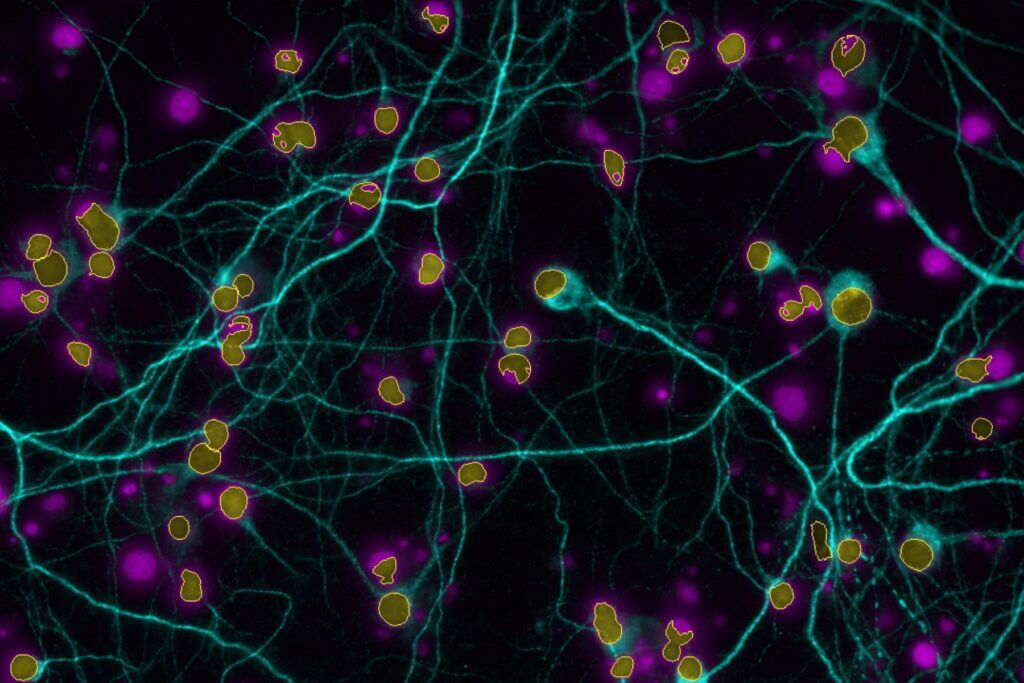
High-Content Imaging
Brain‑on‑Chip platform

Small and Large Animal Facilities
Preclinical Medical Imaging

Immunohistochemistry, Immunofluorescence, and Histology
Immunoassays
The effects of your molecules can be evaluated through complementary and multi-scale approaches.