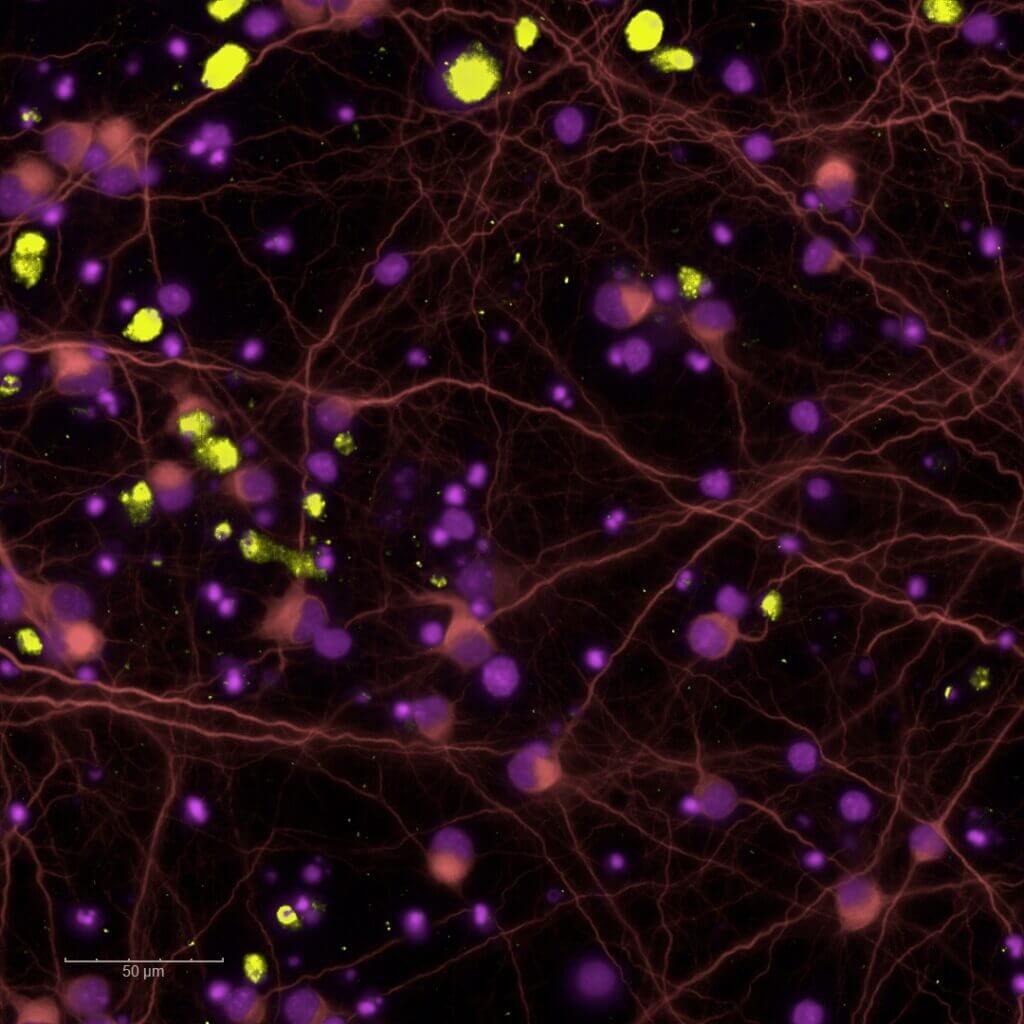
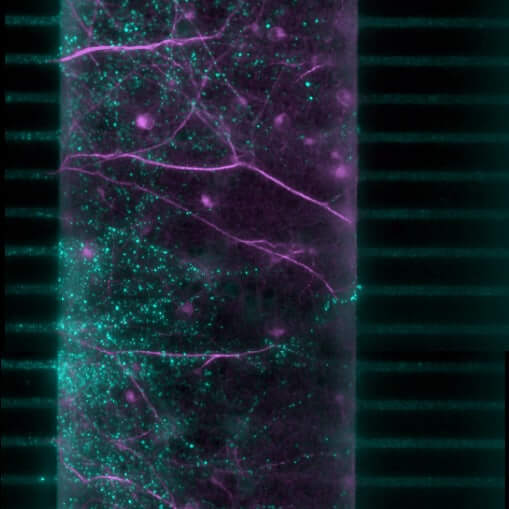
in vitro pharmacology models in Neurology & the CNS
Accelerate the development of your Central Nervous System (CNS) therapies with our in vitro pharmacology models. From hit-to-lead steps to lead characterization in complex cellular systems, our platforms enable you to:
-> Screen and identify neuroprotective, antiaggregating, and thrombolytic candidate compounds;
-> Analyze their mechanisms of action on specific targets, including proteinopathies, neuronal populations, and glial cells;
-> Enhance clinical translatability through the use of brain-on-chip (NAMs) technology.
Aggregation and neutralization assays
The use of our human oligomers provides the opportunity to assess the ability of your candidates to modulate their aggregation kinetics or to specifically interfere with these protein sub‑species. This approach helps you select your compounds with the highest translational potential. Our platform enables you to directly evaluate your candidates on these key pathological targets involved in proteinopathies.
Aggregation assays
Proteinopathies are characterized by the abnormal accumulation of misfolded proteins that assemble into neurotoxic oligomers. Assess the antiaggregating potential of your candidate compounds in a cell-free assay targeting these oligomeric species. This fluorescence-based protocol (ANS and Thioflavin T) monitors aggregation kinetics and the formation of pathogenic oligomeric species using spectrofluorimetry.
The following preparations available for these assays: amyloid β(1-42), Tau, α-synuclein.
Aggregation of human-amyloid β(1-42)
Target the aggregation of human‑amyloid β(1-42), a key pathological driver in Alzheimer’s disease, using our oligomers produced from synthetic, nonrecombinant, endotoxin-free amyloid-β peptide.
Aggregation of human Tau
Target the aggregation of the Tau protein, a key pathological driver in tauopathies, using our oligomers produced from recombinant human Tau protein (2N4R isoform) with a controlled endotoxin level (<1 EU/mL).
Aggregation of human α-synuclein
Target the aggregation of α-synuclein, a key pathological driver in Parkinson’s disease and Lewy body disorders, using our oligomers produced from recombinant human α-synuclein with a controlled endotoxin level (<1 EU/mL).
Detection and recognition of oligomers
Proteinopathies are characterized by the abnormal accumulation of misfolded proteins that assemble into neurotoxic oligomers. Evaluate the neutralization potential of your candidate compounds in a cell-free assay targeting these oligomeric species. This immunoassay-based (Dot-blot) protocol characterizes the recognition of oligomeric aggregates by your candidates and offers a comprehensive assessment of their efficacy and mechanism of action.
The following preparations available for these assays: amyloid β(1-42), Tau, α-synuclein.
Human amyloid-β(1-42)
Target the neutralization of amyloid-β(1-42), a key pathological driver in Alzheimer’s disease, using our oligomers produced from synthetic, nonrecombinant, endotoxin-free amyloid-β peptide.
Human Tau
Target the neutralization of the Tau protein, a key pathological driver in tauopathies, using our oligomers produced from recombinant human Tau protein (2N4R isoform) with a controlled endotoxin level (<1 EU/mL).
Human α-synuclein
Target the neutralization of α-synuclein, a key pathological driver in Parkinson’s disease and Lewy body disorders, using our oligomers produced from recombinant human α-synuclein with a controlled endotoxin level (<1 EU/mL).
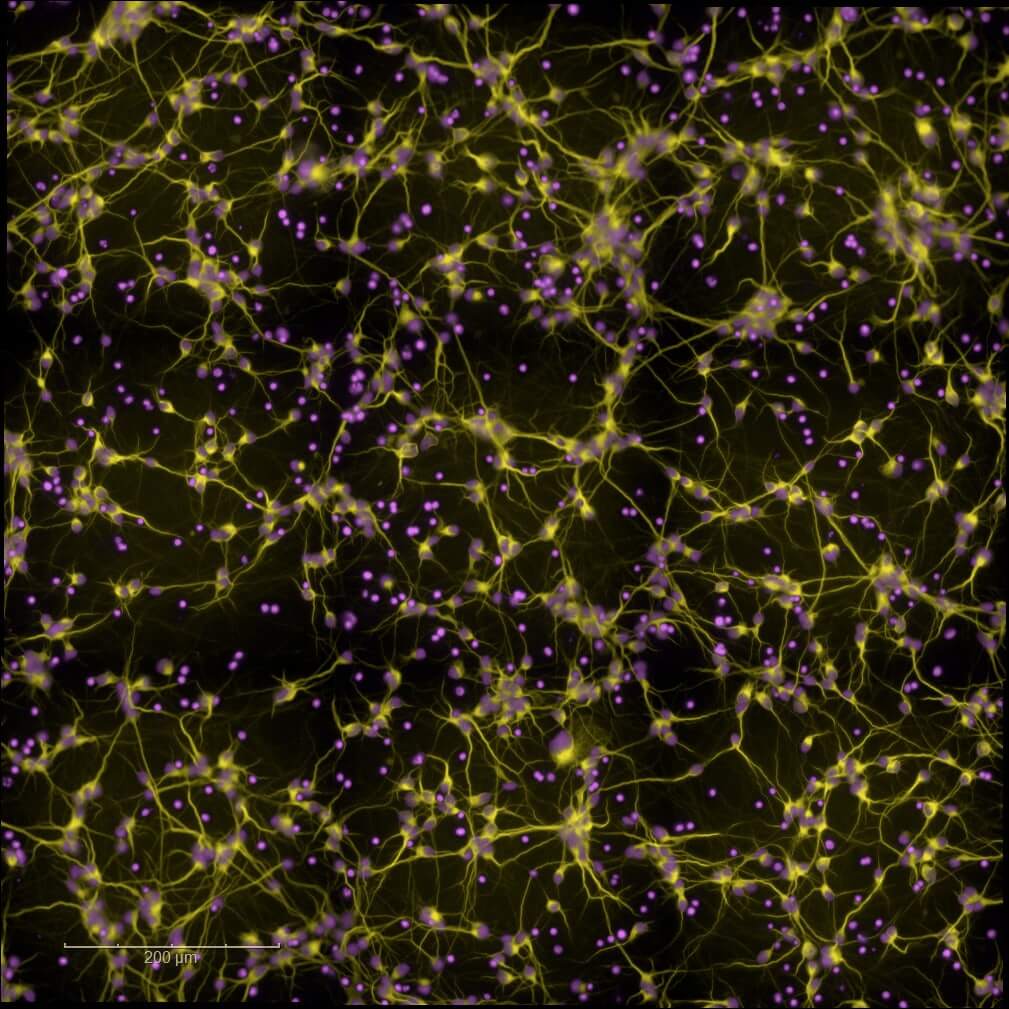
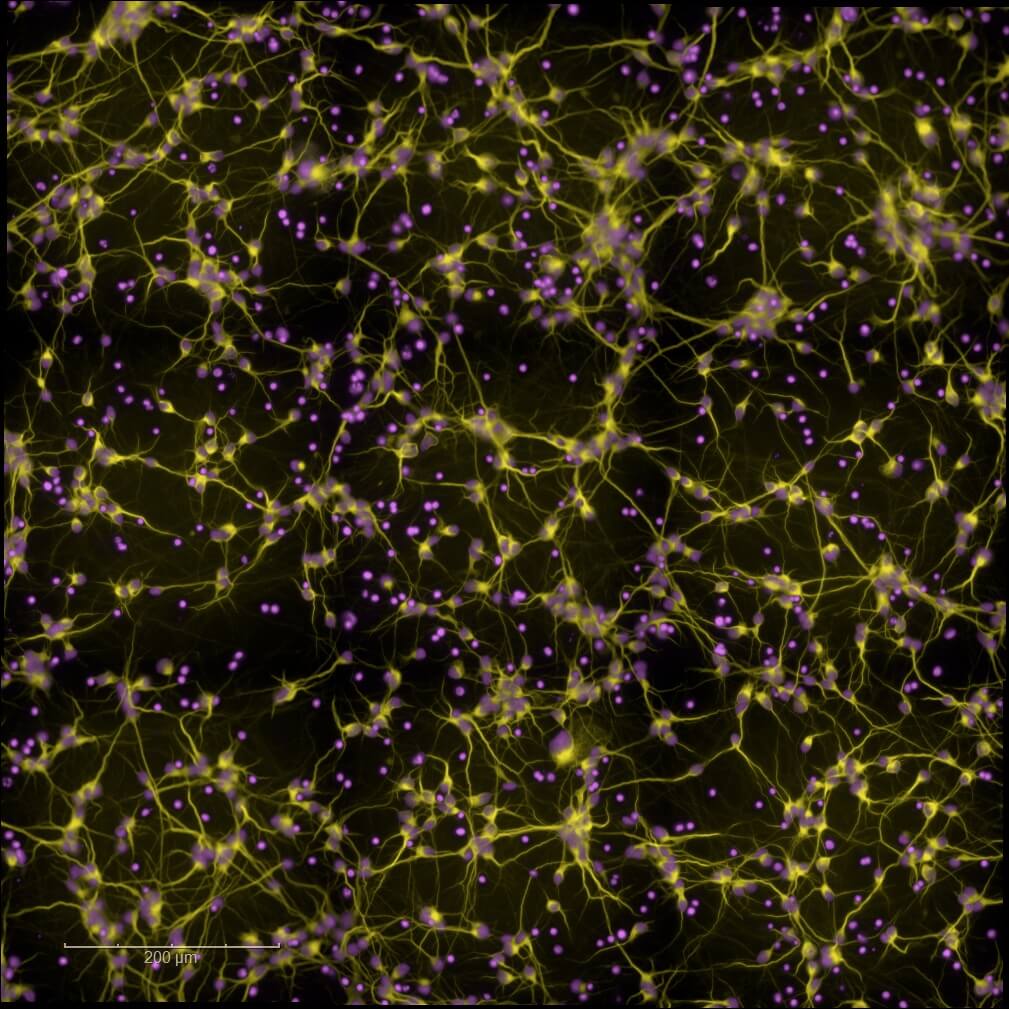
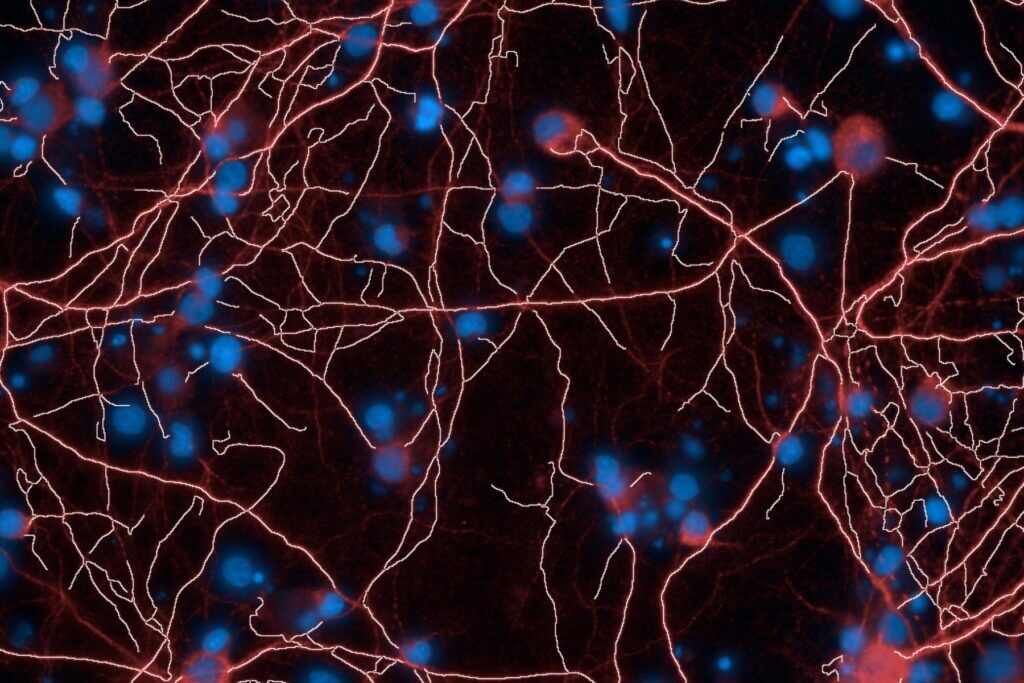
Primary and Human (hiPSC-derived) cells
Validate and optimize your leads with our high-content screening (High Content Imaging) platforms. Leverage our rodent primary cell models or human neuronal and glial cell models (hiPSCs), together with multiparametric analyses, to support candidate selection and guide informed progression toward advanced preclinical development.
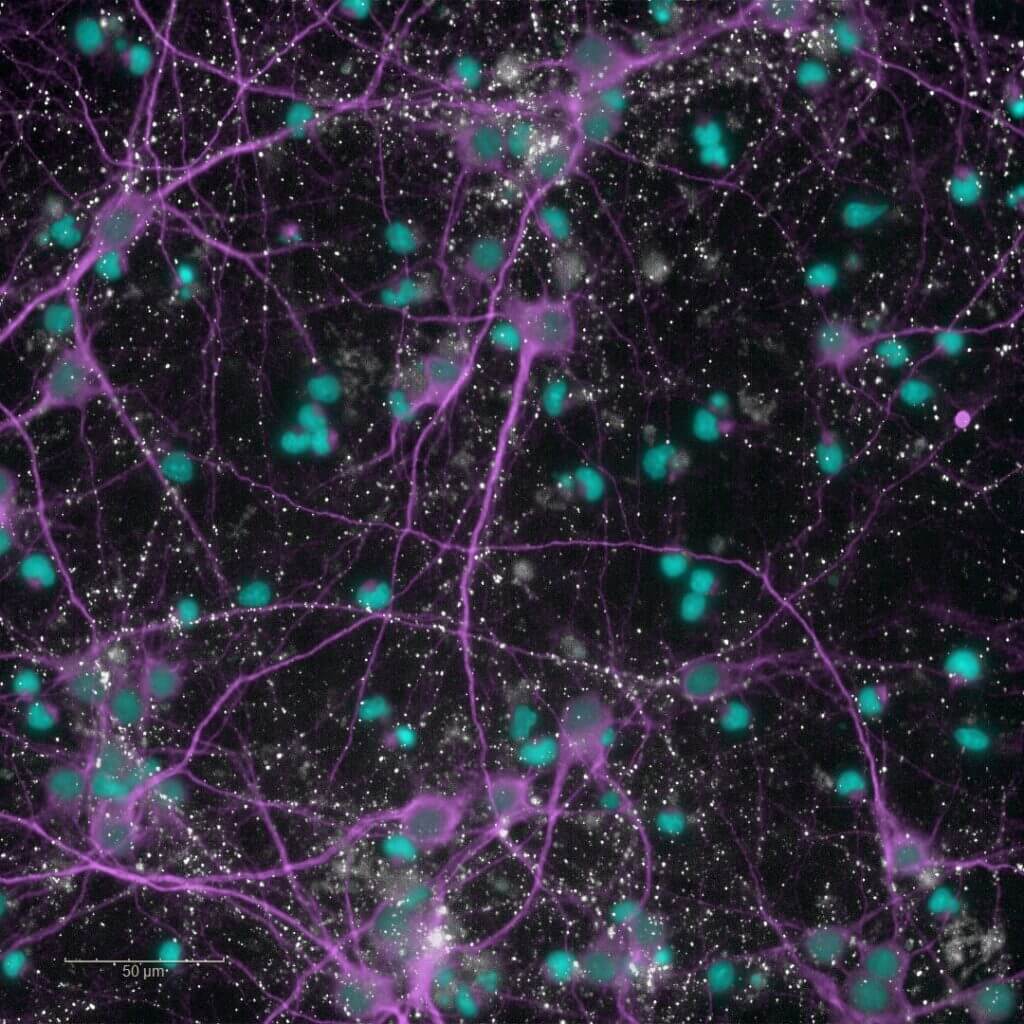
Alzheimer’s disease
Human-amyloid β(1-42) oligomers-induced cytotoxicity
The efficacy of your candidates is rapidly assessed using this cellular model of Alzheimer’s disease. This primary screening assay is based on the exposure of rodent primary cortical neurons to soluble human‑amyloid β(1-42)oligomers. Neuronal viability is quantified using a metabolic activity assay, generating the data required to identify, optimize, and prioritize lead candidates for subsequent advanced mechanistic studies.
Human Tau oligomers-induced cytotoxicity
The efficacy of your candidates is rapidly assessed using this cellular model of tauopathies. This primary screening assay is based on the exposure of rodent primary cortical neurons to soluble human Tauoligomers. Neuronal viability is quantified using a metabolic activity assay, generating the data required to identify, optimize, and prioritize lead candidates for subsequent advanced mechanistic studies.
Human amyloid β(1-42) oligomers-induced neurodegeneration
Beyond neuronal viability, this model— based on rodent primary cortical neurons — recapitulates the neurotoxic cascade of Alzheimer’s disease induced by soluble human amyloid β(1-42) oligomers and quantifies early synaptic degeneration, progressive neurite loss, and final neuronal death. Multiparametric analysis generates the data required to elucidate the mechanism of action and the disease-modifying potential of your candidates.
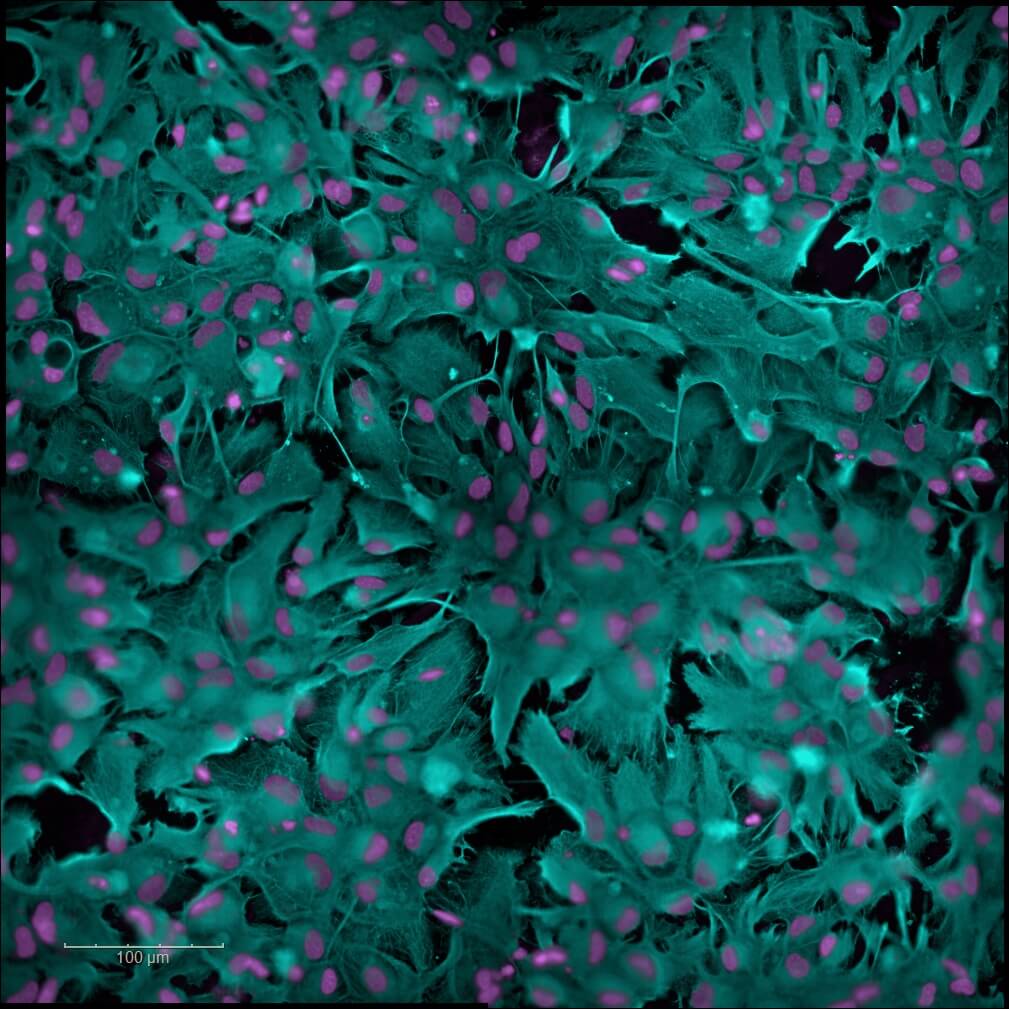
Neuroinflammation
Neuroinflammation is a pathological mechanism common to several neurological disorders, including neurodegenerative diseases. Our human iPSC‑derived astrocyte model enables the evaluation of the anti‑inflammatory properties of your candidates. The pathological neuroinflammatory state is induced either by applying a cocktail of pro‑inflammatory cytokines (TNF‑α, IL‑1β) or by exposing the cells to pathological oligomers. Multiparametric analysis quantifies the modulation of astrogliosis and identifies the candidates capable of restoring a physiological glial environment — a promising strategy for the development of therapies targeting neurodegenerative diseases and CNS pathologies.
Glutamate-induced toxicity : 2D culture
Our 2D excitotoxicity model provides a rapid and reliable platform to screen neuroprotective compounds in primary rodent neuronal cultures exposed to glutamate overload, with MK801 as a reference compound.
Our high‑content imaging platform supports medium‑throughput screening of glutamate‑induced effects at both acute excitotoxic concentrations and sub-toxic levels that reduce synaptic density or disrupt neuritic network organisation.
Excitotoxicity is a key pathophysiological mechanism within the ischemic cascade following stroke. It emerges within minutes to hours of stroke onset and persists throughout post‑ischemic phases (inflammation, synaptic remodelling, and epileptogenic hyperexcitability).
Glutamate-induced toxicity: organ-on-chip
Excitotoxicity is a key pathophysiological mechanism involved in numerous neurological disorders, including neurodegenerative diseases.
Our brain-on-chip model is designed to reproduce excitotoxicity by delivering glutamate specifically into a synaptic chamber located at the interface between two spatially separated neuronal populations (i.e., each cultured in its own independent microphysiological environment). Our microfluidic platform, combined with high-content imaging, enables medium-throughput screening of the effects of your candidate compounds on the isolated synapse, located at a physically distance from the soma and exposed to excessive glutamate.
Glutamate uptake by astrocytes
Astrocytes play a key role in maintaining synaptic homeostasis and preventing excitotoxicity by clearing excess extracellular glutamate. This protective mechanism becomes impaired in reactive astrocytes during neuroinflammatory conditions, particularly in neurodegenerative diseases.
Our astrogliosis model enables the screening of your candidate compounds targeting glutamate uptake in human iPSC-derived astrocytes activated with a cytokine cocktail. Activation of the NFκB pathway is also available as a standard readout through our high-content imaging platform.
Parkinson’s disease
Human α-synuclein oligomers-induced cytotoxicity
The efficacy of your candidates is rapidly assessed using this cellular model of Parkinson’s disease and Lewy body disorders. This primary screening assay is based on the exposure of rodent primary cortical neurons to soluble human α-synuclein oligomers. Neuronal viability is quantified using a metabolic activity assay, generating the data required to identify, optimize, and prioritize lead candidates for subsequent advanced mechanistic studies.
Human α-synuclein oligomers-induced dopaminergic neurodegeneration
Our model, based on rodent primary mesencephalic neurons enriched in dopaminergic neurons and exposed to human α-synuclein oligomers, specifically evaluates the progressive degeneration of this key neuronal population. The multiparametric analysis provides the data required to reveal the disease-modifying potential of your candidates in synucleinopathies.
6-OHDA-induced dopaminergic neurodegeneration
Our model, which uses rodent primary mesencephalic neurons exposed to 6-hydroxydopamine (6-OHDA), specifically targets dopaminergic neurons and mimics toxic oxidative stress. This assay enables the evaluation of the neuroprotective properties of your candidates to counteract this key mechanism of Parkinson’s disease.
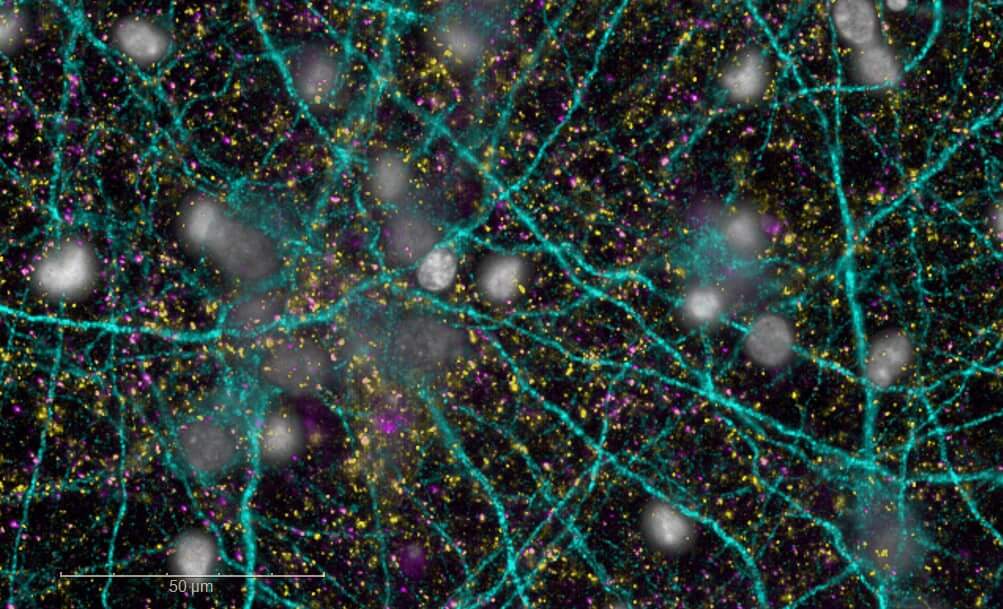
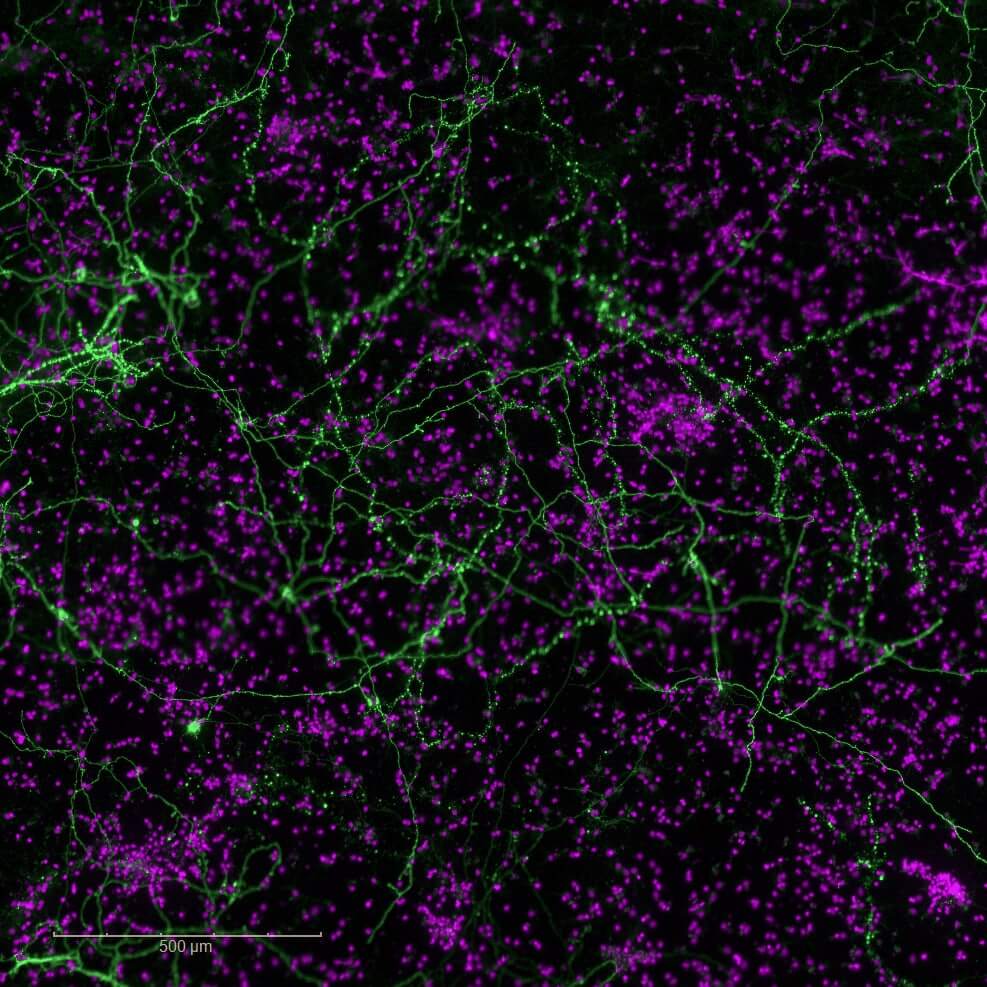
Neuroplasticity models
Synaptogenesis and neuritogenesis
Our model directly evaluates synaptogenesis (formation of new synapses) and neuritogenesis (growth and branching of neurites) in rodent primary cortical neurons. By targeting these fundamental mechanisms of plasticity, identify promising candidates capable of promoting the regeneration of neuronal circuits, counteracting the synaptic disconnection and neuronal loss characteristic of neurodegenerative disorders.
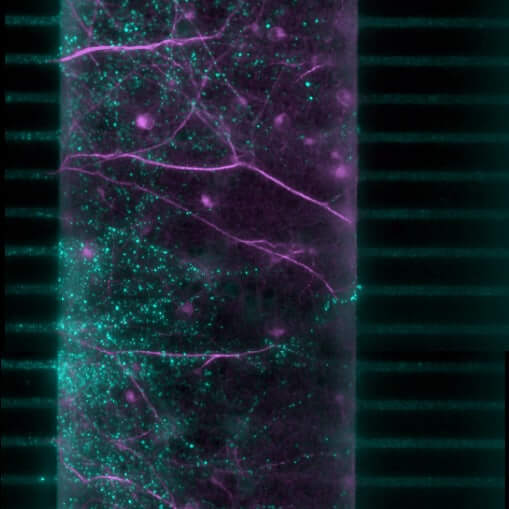
Synaptic plasticity in Brain-on-Chip models
Our brain-on-chip model specifically targets the synaptic compartment to directly quantify synaptogenesis (formation of new synapses) and neuritogenesis (growth and branching of neurites) in rodent primary cortical neurons. By focusing on these fundamental mechanisms of plasticity, identify promising candidates capable of promoting the regeneration of neuronal circuits, counteracting the synaptic disconnection and neuronal loss characteristic of neurodegenerative disorders.
Neuroinflammation models
Alzheimer’s disease
Neuroinflammation is a pathological mechanism common to several neurological disorders, including neurodegenerative diseases. Our human iPSC‑derived astrocyte model enables the evaluation of the anti‑inflammatory properties of your candidates. The pathological neuroinflammatory state is induced by exposing the cells to pathological oligomers. Multiparametric analysis quantifies the modulation of astrogliosis and identifies the candidates capable of restoring a physiological glial environment — a promising strategy for the development of therapies targeting neurodegenerative diseases and CNS pathologies.
Cytokines
Neuroinflammation is a pathological mechanism common to several neurological disorders, including neurodegenerative diseases. Our human iPSC‑derived astrocyte model enables the evaluation of the anti‑inflammatory properties of your candidates. The pathological neuroinflammatory state is induced either by applying a cocktail of pro‑inflammatory cytokines (TNF‑α, IL‑1β). Multiparametric analysis quantifies the modulation of astrogliosis and identifies the candidates capable of restoring a physiological glial environment — a promising strategy for the development of therapies targeting neurodegenerative diseases and CNS pathologies.
Stroke models
Explore our in vitro stroke models for testing your drug candidates on human blood. Find all the details on our page dedicated to our preclinical services for neurovascular diseases.
Glutamate-induced toxicity: 2D culture
Our 2D excitotoxicity model provides a rapid and reliable platform to screen neuroprotective candidate compounds in rodent primary cortical neurons exposed to glutamate overload, with MK801 used as a reference compound.
Our high‑content imaging platform supports medium‑throughput screening of glutamate‑induced effects at non‑cytotoxic concentrations that reduce synaptic density or alter the neuritic network.
Excitotoxicity is a key pathophysiological mechanism involved in numerous neurological disorders, including neurodegenerative diseases.
Glutamate-induced toxicity: organ-on-chip
Excitotoxicity is a key pathophysiological mechanism involved in numerous neurological disorders, including neurodegenerative diseases.
Our brain-on-chip model is designed to reproduce excitotoxicity by delivering glutamate specifically into a synaptic chamber located at the interface between two spatially separated neuronal populations (i.e., each cultured in its own independent microphysiological environment).
Our microfluidic platform, combined with high-content imaging, enables medium-throughput screening of the effects of your candidate compounds on the isolated synapse, located at a physically distance from the soma and exposed to excessive glutamate.
Glutamate uptake by astrocytes
Astrocytes play a key role in maintaining synaptic homeostasis and preventing excitotoxicity by clearing excess extracellular glutamate. This protective mechanism becomes impaired in reactive astrocytes during neuroinflammatory conditions, particularly in neurodegenerative diseases.
Our astrogliosis model enables the screening of your candidate compounds targeting glutamate uptake in human iPSC-derived astrocytes activated with a cytokine cocktail. Activation of the NFκB pathway is also available as a standard readout through our high-content imaging platform.
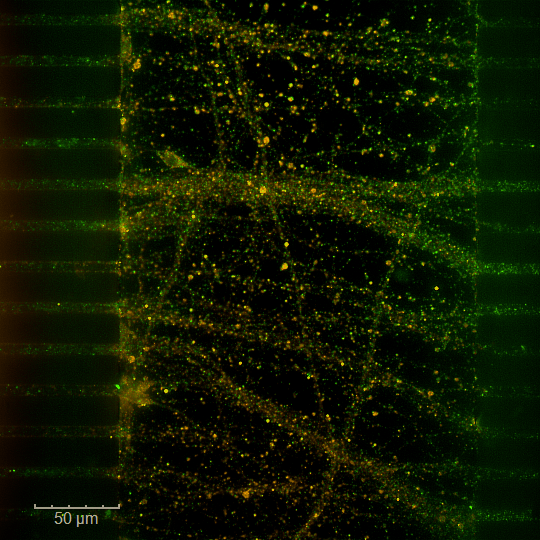
Brain-on-chip – NAMs
Discover new methodological approaches (NAMs) for optimized research within a complex and integrated system thanks to our Organ-on-chip (OOC) technology. Our brain-on-chip (BoC) platform reproduces neuronal architecture and cellular interactions through a compartmentalized microfluidic system. It is the ideal tool to confirm the potential of your candidate compounds by specifically targeting key mechanisms, enabling a predictive evaluation of their efficacy against neurodegeneration and the propagation of proteinopathies.
Take your modeling capabilities to the next level for your most demanding projects.
Neuroplasticity
Synaptic plasticity in Brain-on-Chip models
Our brain-on-chip model specifically targets the synaptic compartment to directly quantify synaptogenesis (formation of new synapses) and neuritogenesis (growth and branching of neurites) in rodent primary cortical neurons. By focusing on these fundamental mechanisms of plasticity, identify promising candidates capable of promoting the regeneration of neuronal circuits, counteracting the synaptic disconnection and neuronal loss characteristic of neurodegenerative disorders.
Excitotoxicity
Glutamate-induced toxicity: organ-on-chip
Excitotoxicity is a key pathophysiological mechanism involved in numerous neurological disorders, including neurodegenerative diseases.
Our brain-on-chip model is designed to reproduce excitotoxicity by delivering glutamate specifically into a synaptic chamber located at the interface between two spatially separated neuronal populations (i.e., each cultured in its own independent microphysiological environment). Our microfluidic platform, combined with high-content imaging, enables medium-throughput screening of the effects of your candidate compounds on the isolated synapse, located at a physically distance from the soma and exposed to excessive glutamate.
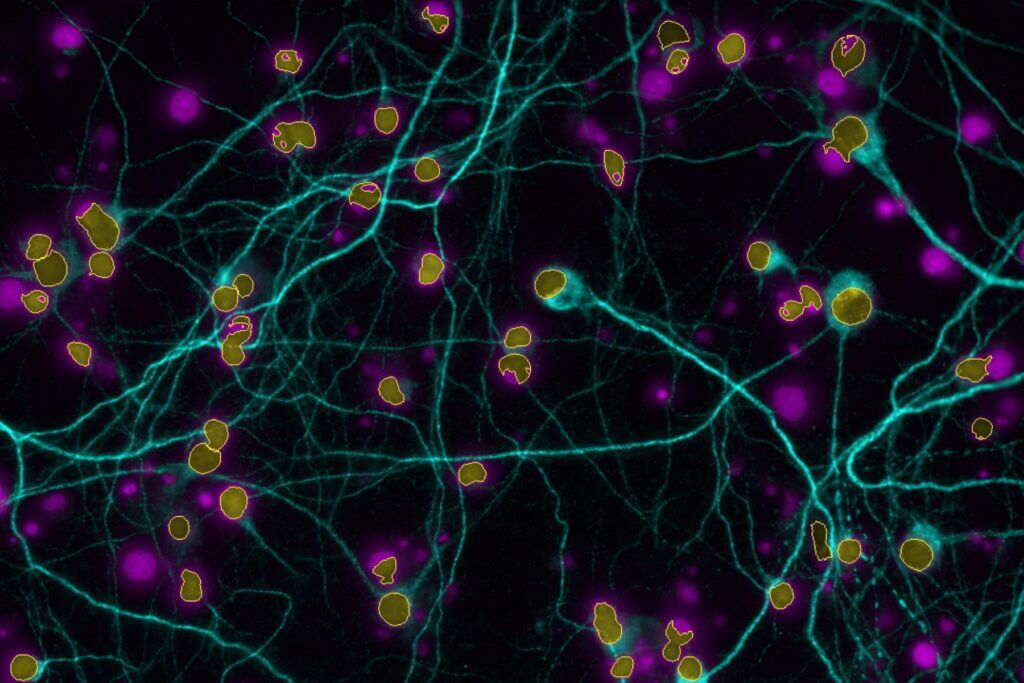
High-Content Imaging
Brain‑on‑Chip platform

Small and Large Animal Facilities
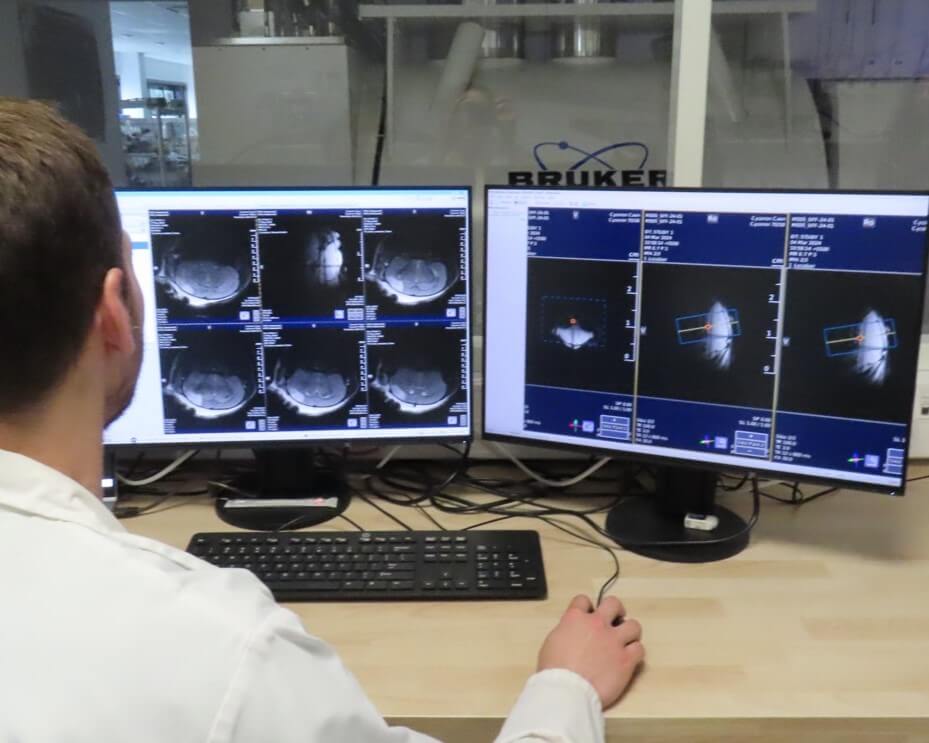
Preclinical Medical Imaging

Immunohistochemistry, Immunofluorescence, and Histology
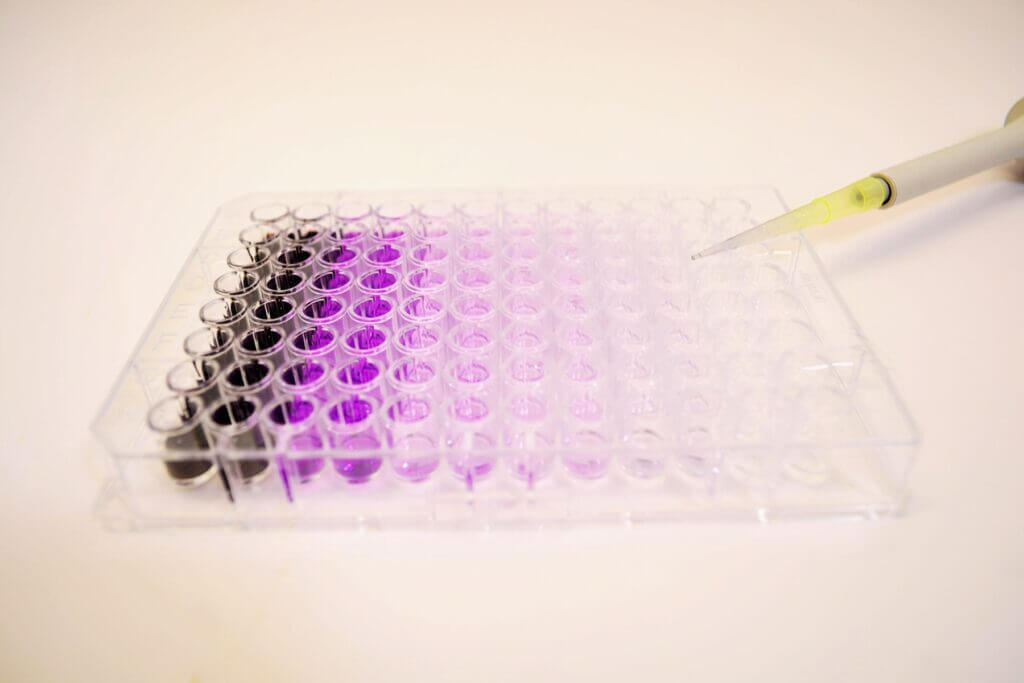
Immunoassays
The effects of your molecules can be evaluated through complementary and multi-scale approaches.