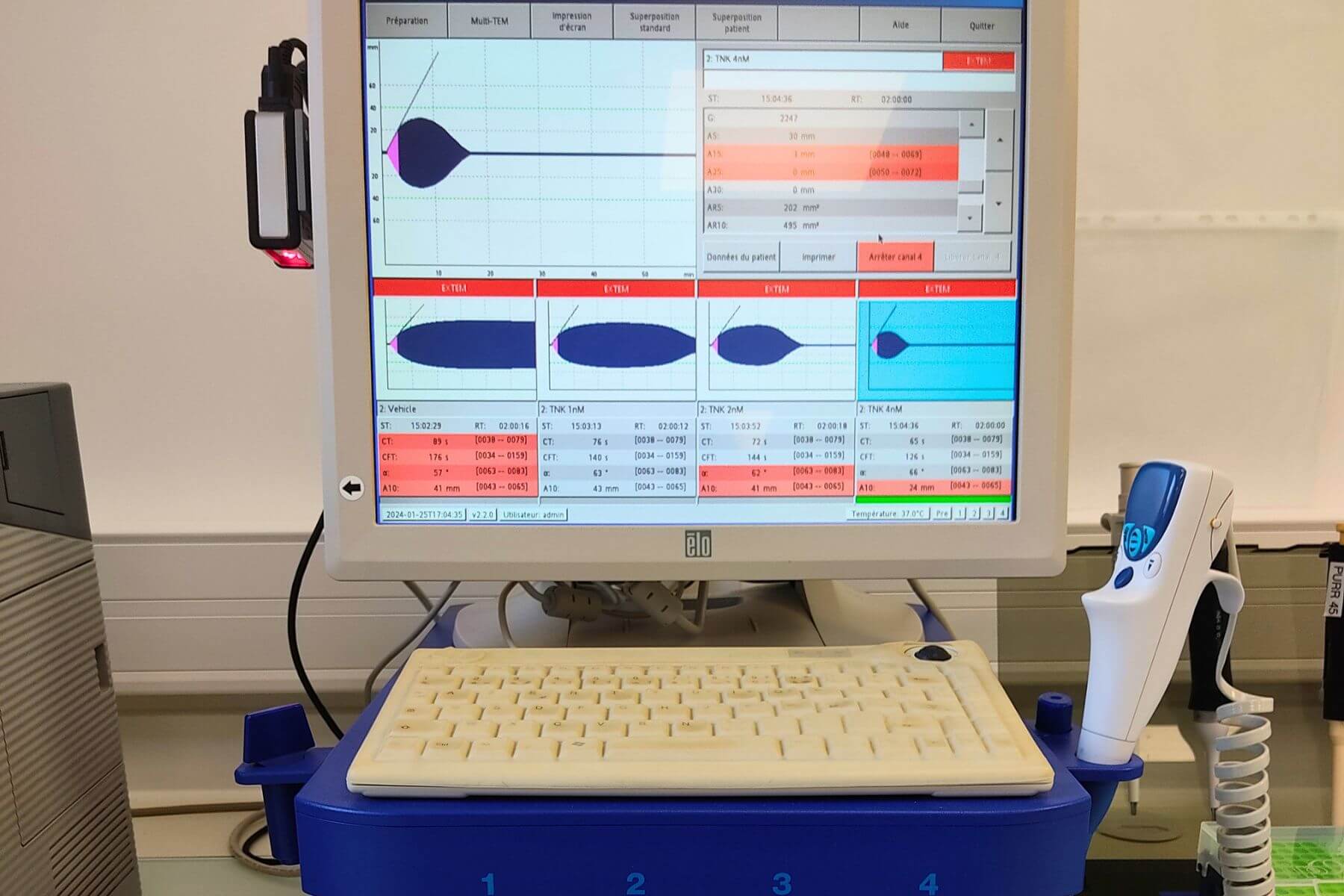
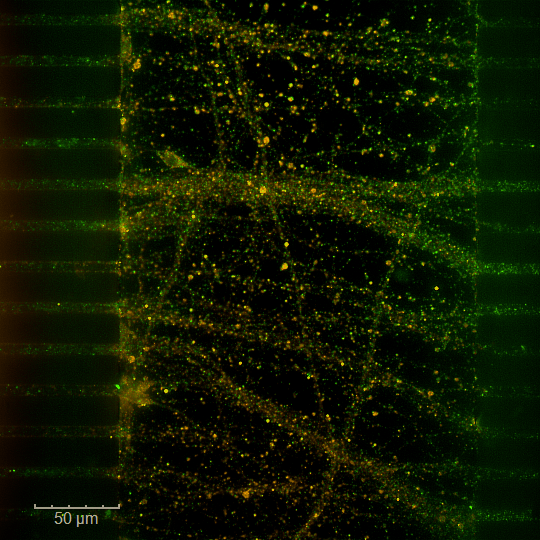
Injecting sizecalibrated autologous blood clots induces embolic occlusion of the proximal MCA segment. This model produces cortical and subcortical lesions similar to those observed in mechanical ischemia models. Mortality, deficit severity, and responsiveness to the reference fibrinolytic treatment (Alteplase) can be modulated by adjusting the amount of thrombotic material used for the embolic event. This original and wellcharacterized model enables the evaluation of a wide range of therapeutic strategies, together with longterm monitoring of functional recovery.
Validated model available in mice and rats.