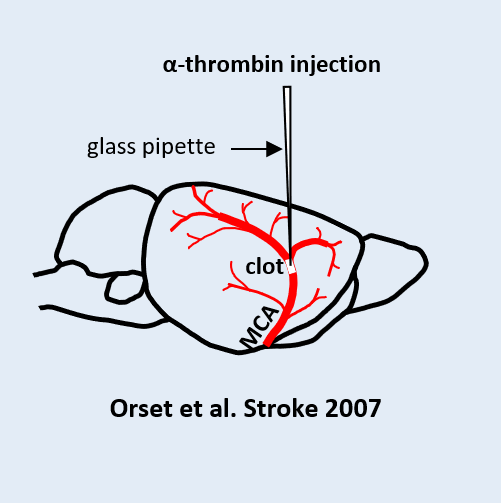
The pulmonary embolism model induced by injection of autologous clots relies on preparing and injecting pre‑formed calibrated clots into the jugular vein to selectively obstruct the pulmonary arteries. This model reproduces the acute increase in pulmonary pressure observed in human clinical settings, along with significant stress on the right ventricle and secondary vascular remodeling. The severity of the condition can be adjusted based on the amount of thrombotic material used. Its strong reproducibility and rapid implementation make it suitable for quickly evaluating the efficacy of thrombolytic, vasodilatory, or cardioprotective treatments.
Rat model.